Can a displacement reaction be a redox reaction justify?
.
Also know, can a double displacement reaction be a redox reaction?
Redox reactions can be displacement reactions. For example, Zn displaces copper from copper sulphate solution is a redox reaction, in which Zinc is oxidized and copper is reduced. Double displacement reactions are not redox reactions, as they do not involve change in oxidation number of either of the reactant.
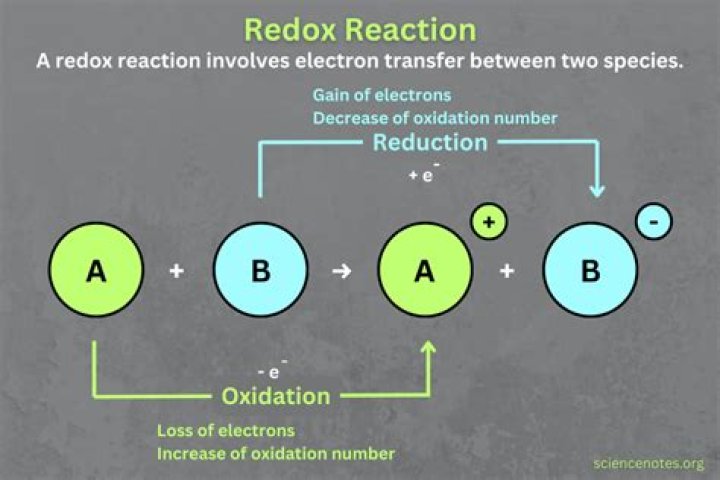
Also Know, what makes a redox reaction? Oxidation-reduction reactions (redox reactions) are reactions in which electrons are lost by an atom or ion in one reactant and gained by an atom or ion in another reactant. When reduction occurs, an element gains electrons and its oxidation number decreases or is reduced (becomes more negative).
In this way, what happens in a displacement reaction justify your answer Giving one example?
Displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound. Both metals and non-metals take part in displacement reactions. Example : Reaction of iron nails with copper sulphate solution.
What is the difference between displacement and double displacement reaction?
In a displacement reaction, a more reactive element replaces a less reactive element from a compound. In a double displacement reaction, two atoms or a group of atoms switch places form new compounds. In a displacement reaction, one displacement takes place. Salt solutions of two different metals react with each other.
Related Question AnswersWhat are the types of redox reactions?
Keep this in mind as we look at the five main types of redox reactions: combination, decomposition, displacement, combustion, and disproportion.- Combination. Combination reactions “combine” elements to form a chemical compound.
- Decomposition.
- Displacement.
- Combustion.
- Disproportionation.
Are single displacement reactions redox?
Single Displacement Reactions. Another type of redox reaction is one in which an element replaces or displaces another from a compound. In these reactions, known as single replacement reactions, the element which replaces that which is in a compound is always oxidized. The element being displaced, is always reduced.Are double displacement reactions reversible?
Answer: In a irreversible reaction, the reactants react to form the products, which cannot revert back into reactants. When a double displacement reaction occurs, the cations and anions switch partners, resulting in the formation of two new ionic compounds AD and CB, one of which is in the solid state.Are all double displacement reactions precipitation reactions?
It is not necessary for all double displacement reaction to be precipitation reaction because there are several such reaction in which one reactant is acid and other one is base. The acid reacts with base and form salt which is found dissolved in water that is formed.Is agno3 NaCl a redox reaction?
IS NaCl+AgNO3-AgCl+NaNO3 A REDOX REACTION? No Because nothing has been oxidised or reduced.Which reactions are not redox reactions?
Examples of non-redox reaction;- Neutralization reaction.
- Double decomposition reaction.
Is neutralization a redox reaction?
In chemistry, neutralization is a chemical reaction in which an acid and a base react to form a salt. Redox (or reduction-oxidation) reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species.Can a displacement reaction be a redox reaction explain with the help of an example?
Yes, a simple displacement reaction is also a redox reaction as it is always accompanied by oxidation-reduction. In this reaction, the more electronegative chlorine displaces iodine from its salt, potassium iodide. Here, zinc is oxidised to +2 state from zero state, and hydrogen is reduced to zero state from +1 state.What is displacement reaction explain with example?
Displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound. Both metals and non-metals take part in displacement reactions. Example : Reaction of iron nails with copper sulphate solution.How do you know if a displacement reaction has occurred?
what are three ways to recognize if a single displacement reaction has taken place?- Color change.
- Formation of bubbles (a gas)
- Formation of a precipitate.