Only Aldehydes containing alpha Hydrogen ( acidic Hydrogen ) give positive results with Fehling's Test (Formaldehyde also gives positive results ). This is because Rate-Determining step of the reaction is the formation of enolate ..
Consequently, does formaldehyde give tollens test?
Note-formaldehyde does not give Tollen's test.
Also Know, which aldehyde does not give Fehling's test? Aldehydes that lack alpha hydrogens, such as benzaldehyde or pivalaldehyde (2,2-dimethylpropanal) cannot form an enolate and thus do not give a positive Fehling's test result under usual conditions.
Besides, what happens when formaldehyde is heated with Fehling solution?
Hydrolysis of trichloroacetone gives chloroform. (iii) Formaldehyde reacts with Fehling Solution to form anion of formic acid.
How can you tell the difference between formaldehyde and benzaldehyde?
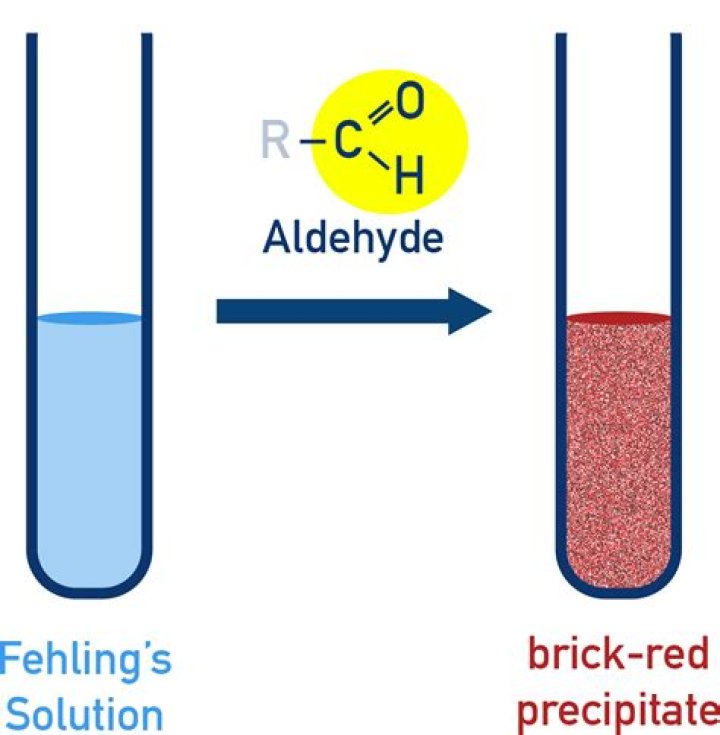
Benzaldehyde and formaldehyde can be distinguished by Fehling's test. Fehling's solution is an alkaline solution of copper sulphate containing sodium potassium tartarate. When fehling's solution will be added to formaldehyde , methanoic acid and cuprous oxide (red precipitate ) will be formed.
Related Question Answers
Does formaldehyde give silver mirror test?
ANSWER: Yes, formaldehyde give silver mirror test. EXPLANATION: Silver mirror test is the tollen's reagent.Which is more reactive formaldehyde and benzaldehyde?
where as in formaldehyde the -h groups attached to the carbonyl carbon get a highly positive change and thus more reactive than that of benzaldehyde. Formaldehyde has a H- (Hydrogen) attached to the Carbonyl carbon of -CHO (aldehyde) group. Whereas , Benzaldehyde has a Ph- (Phenyl) group attached.What gives a positive tollens test?
Tollens' test: A chemical reaction used to test for the presence of an aldehyde or a terminal α-hydroxy ketone. A terminal α-hydroxy ketone gives a positive Tollens' test because Tollens' reagent oxidizes the α-hydroxy ketone to an aldehyde.Why do ketones not react with Fehling solution?
Fehling's can be used to determine whether a carbonyl-containing compound is an aldehyde or a ketone. The compound to be tested is added to the Fehling's solution and the mixture is heated. Aldehydes are oxidized, giving a positive result, but ketones do not react, unless they are alpha-hydroxy-ketones.Why does benzaldehyde not respond to Fehling's test?
In Fehling test, enolate formation takes place, thus Aldehydes that lack alpha hydrogen cannot form an enolate and thus do not give a positive Fehling's test. Thus Benzaldehyde do not give Fehling test as it do not have α− Hydrogen. While Acetaldehyde have 3α−Hydrogen thus it can form enolate and undergo Fehling test.Does formaldehyde give iodoform test?
Formaldehyde and acetaldehyde can be distinguished by iodoform test. - Methyl ketones react with iodine and potassium hydroxide to give yellow precipitate. - Formaldehyde doesn't give iodoform test.Why does formic acid give tollens test?
Formic acid (HCOOH) reduces Tollen's reagent. It contains a hydrogen atom instead of an alkyl group. Like aldehydes it can be easily oxidised to carbonic acid (H2CO3) which decomposes to give CO2 and H2O. Thus like aldehydes it reduces Tollen's reagent to silver mirror.What compounds give Fehling tests?
Formic acid (i.e HCOOH - methanoic acid) is a compound which gives a positive test with Fehling's,as well as with Tollens' test. This is due to the fact because it is readily oxidizable to carbon dioxide and water.Which sugar does not reduce Fehling's solution?
Sucrose does not react with Fehling's solution, as this disaccharide of fructose and glucose (2,1 glycosidic linkage) has no free aldehyde or ketone groups.What is Fehling reagent formula?
The deep blue active ingredient in Fehling's solution is the bis(tartrate) complex of Cu 2+. The net reaction between an aldehyde and the copper(II) ions in Fehling's solution may be written as: RCHO + 2 Cu 2+ + 5 OH − → RCOO − + Cu 2O + 3 H 2O.What is Fehling A and B?
Fehling's A is aqueous solution of copper(II) sulfate, which is deep blue. Fehling's B is a colorless solution of aqueous potassium sodium tartrate (also known as Rochelle salt) made in a strong alkali, commonly with sodium hydroxide. Typically, the L-tartrate salt is used.How do you make Fehling's?
Two solutions are required: Fehling's "A" uses 7 g CuSO4. 5H2O dissolved in distilled water containing 2 drops of dilute sulfuric acid. Fehling's "B" uses 35g of potassium tartrate and 12g of NaOH in 100 ml of distilled water.What do you mean by Fehling Solution?
Medical Definition of Fehling's solution : a blue solution of Rochelle salt and copper sulfate used as an oxidizing agent in a test for sugars and aldehydes in which the precipitation of a red oxide of copper indicates a positive result.Why is Fehling A separated from Fehling B?
The reason Fehlings Solution A and B are kept separate is because if you combine the two, the Copper Tartarate complex that is formed will quickly degrade, and will not be effective in the detection of “reducing sugars” (sugars containing aldehyde groups).How can Fehling's test determine the reducing property of sugar?
Barfoed's Reagent a mixture of ethanoic (acetic) acid and copper(II) acetate, is added to the test solution and boiled. If any reducing sugars are present a red precipitate of copper(I) oxide is formed withing three minutes. The structure of the copper(II) acetate dimer has been determined.What is Fehling's test used for?
Fehling's solution. Fehling's solution is a chemical reagent used to differentiate between water-soluble carbohydrate and ketone functional groups, and as a test for reducing sugars and non-reducing sugars, supplementary to the Tollens' reagent test. The test was developed by German chemist Hermann von Fehling in 1849.Why is there a need to freshly prepare the Fehling's reagent?
They are prepared whenever needed and not beforehand because this Fehling's solution decomposes with time and becomes non-effective for the tests!Which does not react with Fehling's solution?
Aldehydes are oxidized, giving a positive result, but ketones do not react, unless they are alpha-hydroxy-ketones. A negative result is the absence of the red precipitate; it is important to note that Fehling's will not work with aromaticaldehydes; in this case Tollens' reagent should be used.What is the difference between Fehling's test and Benedict's test?
For this identification, Benedict's test and Fehling's test can be used. The main difference between Benedict's solution and Fehling's solution is that Benedict's solution contains copper(II) citrate whereas Fehling's solution contains copper(II) tartrate.