Does NaCl have ion-dipole forces?
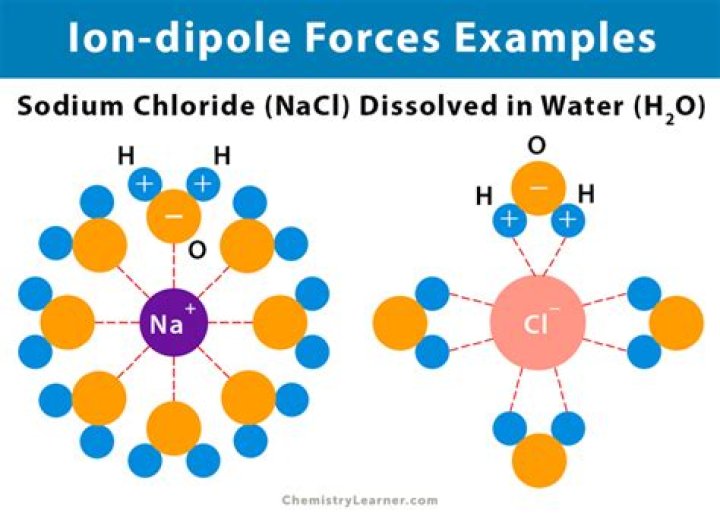
Does NaCl have ion-dipole forces?
Ion-Dipole Forces are involved in solutions where an ionic compound is dissolved into a polar solvent, like that of a solution of table salt (NaCl) in water.
Is NaCl dipole-dipole?
For example, NaCl has the highest dipole moment because it has an ionic bond (i.e. highest charge separation)….Introduction.
| Compound | Dipole Moment (Debyes) |
|---|---|
| NaCl | 9.0 (measured in the gas phase) |
| CH3Cl | 1.87 |
| H2O | 1.85 |
| NH3 | 1.47 |
What kind of intermolecular forces does NaCl have?
Ion – dipole forces – Intermolecular force between an ion and the oppositely charged end of a polar molecule. e.g. NaCl in H2O. 3.
Is NaCl and H2O ion-dipole?
Ion-Dipole: forces of attraction between a cation/anion and the solvent molecules when dissolved in water or other polar molecules; e.g., NaCl dissolving in H2O to form Na+ and Cl- surrounded by water molecules. These are much stronger forces of attraction than other dipole-dipole forces.
Is NaCl an ionic force?
NaCl is an ionic compound because the bond formed between sodium and the chlorine atom is ionic in nature, it is formed due to the transfer of electrons from Na to Cl, which results in the formation of ions(Na+ and Cl–), later these ions are attracted by the electrostatic force of attraction and forms the ionic bond.
Is NaCl an ion ion?
Sodium chloride (NaCl) is a typical ionic compound. Sodium has 1 electron in its outermost shell, and chlorine has 7 electrons. It is easiest for sodium to lose its electron and form a +1 ion, and for chlorine to gain an electron, forming a -1 ion.
What are ion-dipole forces?
An ion-dipole force is an attractive force that results from the electrostatic attraction between an ion and a neutral molecule that has a dipole. Most commonly found in solutions. A negative ion (anion) attracts the partially positive end of a neutral polar molecule.
Is NaCl covalent or ionic?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
Is NaCl a covalent compound?
Covalent Bonding. Ionic compounds, such as sodium chloride (NaCl), are formed by a transfer of electrons that creates ions. The hydrogen and oxygen atoms in a water molecule, however, are bonded by sharing electrons rather than by transferring them.