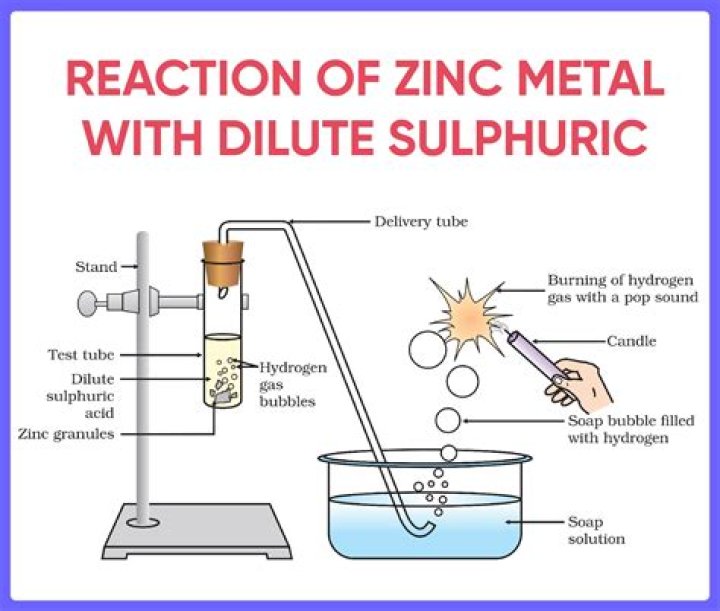
Zinc reacts with sulphuric acid to form zinc sulphate and hydrogen gas is liberated. Zinc + sulphuric acid ——→ zinc sulphate + hydrogen..
Moreover, what happens when zinc reacts with Sulphuric acid?
Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved. This is a single displacement reaction of a non-metal by a metal. The zinc will react with sulphuric acid forming zinc sulphate dissolved in solution and releasing hydrogen gas.
Subsequently, question is, what salt does zinc and sulfuric acid make? Naming salts
| Hydrochloric acid | Sulfuric acid |
| Copper oxide | Copper chloride | Copper sulfate |
| Aluminium hydroxide | Aluminium chloride | Aluminium sulfate |
| Zinc carbonate | Zinc chloride | Zinc sulfate |
Furthermore, does Zinc react with acid?
The metal zinc readily reacts with hydrochloric acid to produce hydrogen gas (H2) and zinc chloride (ZnCl2). The reagents are zinc and hydrochloric acid, and the sum is 0 + 2 * ( -167.2) = -334.3.
What is the balanced equation for zinc and sulfuric acid?
Zn + H2SO4 = ZnSO4 + H2 | Chemical reaction and equation.
Related Question Answers
Why do you react the excess zinc with sulfuric acid?
Zinc is more reactive than hydrogen and it displaces hydrogen from dilute acids. Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved. This is a single displacement reaction of a non-metal by a metal. So, this reaction is a chemical change.What is the Colour of zinc chloride?
white
What is the action of concentrated Sulphuric acid on zinc?
Zinc reacts with sulfuric acid to produce hydrogen. The reaction is catalysed by copper. The rate of reaction can be compared by means of the rate of production of hydrogen gas bubbles.What happens when zinc reacts with nitric acid?
The products formed when zinc reacts with dilute nitric acid are zinc nitrate and hydrogen gas; i.e. 5Zn + 12HNO3 ----> 5Zn(NO3)2 + N2 + 6H2O. These products are formed due to the reduction of HNO3 by nascent hydrogen, which is produced in increasing amounts as the concentration of nitric acid is lowered; e.g.What happens when copper reacts with dilute Sulphuric acid?
Copper does not react with dilute sulphuric acid as its reduction potential is higher than that of hydrogen. Copper does not displace hydrogen from non-oxidising acids like HCl or dilute H2SO4. But, concentrated sulphuric acid is an oxidising agent. The other products of the reaction are copper(II) sulphate and water.What happens when zinc reacts with sodium hydroxide?
Zinc is a transition metal which shows moderate reactivity. Zinc reacts with NaOH to form sodium zincate with the evolution of hydrogen gas.What reacts with dilute sulfuric acid?
Dilute sulfuric acid reacts with metals via a single displacement reaction as with other typical acids, producing hydrogen gas and salts (the metal sulfate). It attacks reactive metals (metals at positions above copper in the reactivity series) such as iron, aluminium, zinc, manganese, magnesium, and nickel.What is the formula of dilute Sulphuric acid?
The formula for sulfuric acid is H2SO4. The molar mass of sulfuric acid is 98.07848 g mol.What acids does Zinc react with?
Zinc reacts with dilute nitric acid to give zinc nitrate and hydrogen gas.What does Zinc react well with?
Zinc reacts readily with acids, alkalis and other non-metals. Extremely pure zinc reacts only slowly at room temperature with acids. Strong acids, such as hydrochloric or sulfuric acid, can remove the passivating layer and subsequent reaction with water releases hydrogen gas.Why does zinc not react with water?
Zinc does not react with water, because it, too, forms a protective layer of insoluble zinc hydroxide, Zn(OH)2 .What happens when you mix hydrochloric acid and zinc?
Discussion: Zinc is oxidized by hydrochloric acid to form zinc chloride. In the process, hydrogen gas is produced. Safety: HCl and zinc chloride are corrosive and can cause skin irritations or burns.How does Zinc react with oxygen?
react with oxygen in the air to form the metal oxide. Zinc reacts fairly quickly to form zinc oxide. zinc + oxygen. zinc oxide.Does Zinc react with bases?
Accepted Answer: Most metals do not react with bases but zinc metal does because it is amphoteric. That is, it reacts with acids as well as bases. When sodium hydroxide solution is heated with zinc, then sodium zincate and hydrogen gas are formed.Does Zinc rust in water?
All zinc galvanized coatings are more corrosion resistant than bare iron or steel. Like all ferrous metals, zinc corrodes when exposed to air and water. However, zinc corrodes at a rate of 1/30 of that for steel. Also like other ferrous metals, zinc corrodes or rusts at different rates depending on its environment (8).Does hydrochloric acid dissolve zinc?
Yes, zinc (Zn) dissolves in hydrochloric acid (HCl). Zinc is more reactive than hydrogen, as the reactivity series says. Therefore, zinc can displace hydrogen from the HCl and form its soluble chloride, that is, zinc chloride (ZnCl2).What is zinc chloride in?
Zinc Chloride is a white crystalline solid. In cosmetics and personal care products, Zinc Chloride is used in the formulation of dentifrices, mouthwashes, skin care products, hair conditioners and bath products.What salt is produced by Sulphuric acid?
Neutralising sulphuric acid produces sulphate salts. Sulphuric acid + sodium hydroxide → sodium sulphate + water.Which is the weakest acid?
The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF). While technically a weak acid, hydrofluoric acid is extremely powerful and highly corrosive.