How does a molten carbonate fuel cell work?
How does a molten carbonate fuel cell work?
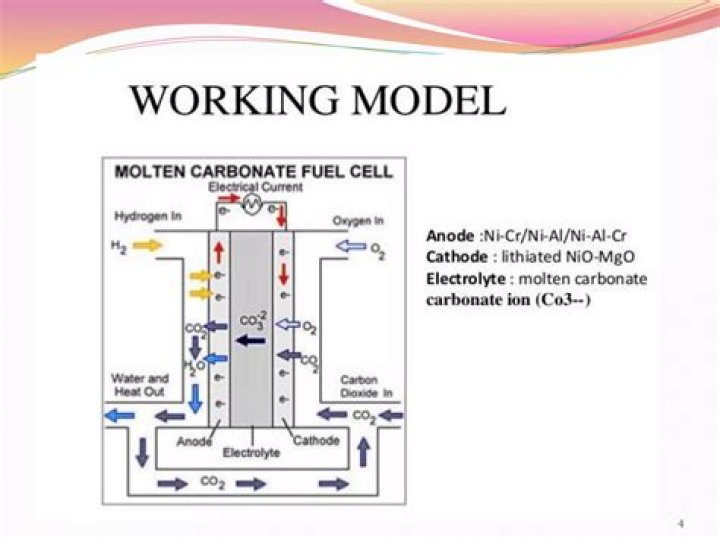
In a molten carbonate fuel cell (MCFC), carbonate salts are the electrolyte. Heated to 650 degrees C (about 1,200 degrees F), the salts melt and conduct carbonate ions (CO3) from the cathode to the anode. At the anode, hydrogen reacts with the ions to produce water, carbon dioxide, and electrons.
What catalyst is used in fuel cells?
platinum catalyst
On the anode side, the platinum catalyst enables hydrogen molecules to be split into protons and electrons. On the cathode side, the platinum catalyst enables oxygen reduction by reacting with the protons generated by the anode, producing water.
Which catalyst is used in solid oxide fuel cell?
Nickel-Based Electrodes. The standard material in SOFC anodes and SOE cathodes based on YSZ electrolytes is a ceramic-metallic (cermet) composite of Ni and YSZ (14), with Ni providing both electronic conductivity and catalytic activity in this application.
What is the disadvantage of molten carbonate fuel cell?
The primary disadvantage of MFCFs is that their high temperatures decrease cell life. There is currently research underway to find corrosion resistant materials for use at high temperatures.
How do methanol fuel cells work?
It has an anode and a cathode. They are separated by a membrane. By means of an electrochemical reaction, the direct methanol fuel cell converts the fuel, i.e. methanol, into electricity in combination with oxygen. A heat engine drives a generator and thus makes the electricity usable.
How do direct methanol fuel cells work?
The DMFC relies upon the oxidation of methanol on a catalyst layer to form carbon dioxide. Water is consumed at the anode and produced at the cathode. Electrons are transported through an external circuit from anode to cathode, providing power to connected devices.
Which catalyst is used as catalyst for fuel cell at anode or cathode?
platinum
Proton exchange membrane fuel cells (PEMFCs) dominate the transportation fuel cell market and platinum (Pt) is the catalyst material used for both anode and cathode.
What is a PEM membrane made of?
A proton-exchange membrane, or polymer-electrolyte membrane (PEM), is a semipermeable membrane generally made from ionomers and designed to conduct protons while acting as an electronic insulator and reactant barrier, e.g. to oxygen and hydrogen gas.
Which electrolyte is used in solid oxide fuel cell?
The most common solid electrolyte material used in solid oxide fuel cells is yttria-stabilized zirconia (YSZ). Yttria is added to stabilize the conductive cubic fluorite phase, as well as to increase the concentration of oxygen vacancies, and thus increase the ionic conductivity.
Which fuel is used in solid oxide fuel cell?
Fuel : PEM fuel cells use pure hydrogen (H2) as fuel. Meanwhile, solid oxide fuel cells can use hydrocarbon fuels such as natural gas, methane and propane to produce electricity.
Which fuel cell has the highest operating temperature Mcq?
Explanation: The Molten Carbon Fuel Cell operates at high temperatures and pressures.
What is used as a catalyst in direct methanol fuel cell?
The need for water limits the energy density of the fuel. Platinum is used as a catalyst for both half-reactions. This contributes to the loss of cell voltage potential, as any methanol that is present in the cathode chamber will oxidize.



