How does plant pigment chromatography work?
.
In this manner, how is pigment extracted from plants?
Procedures
- Obtain a mortar and pestle and choose a plant for your extraction.
- Add the plant to the mortar and then crush your plant in the mortar with the pestle.
- Crush as much as you can and then add the solvent (Water, Acetone and or Ethanol).
- Filter the extract to remove all non-soluble plant material.
Additionally, what does the RF value tell you about plant pigments? The Rf values indicate how soluble the particular pigment is in the solvent by how high the pigment moves on the paper. Two pigments with the same Rf value are likely to be identical molecules. Small Rf values tend to indicate larger, less soluble pigments while the highly soluble pigments have an Rf value near to one.
Simply so, what is paper chromatography and how does it work?
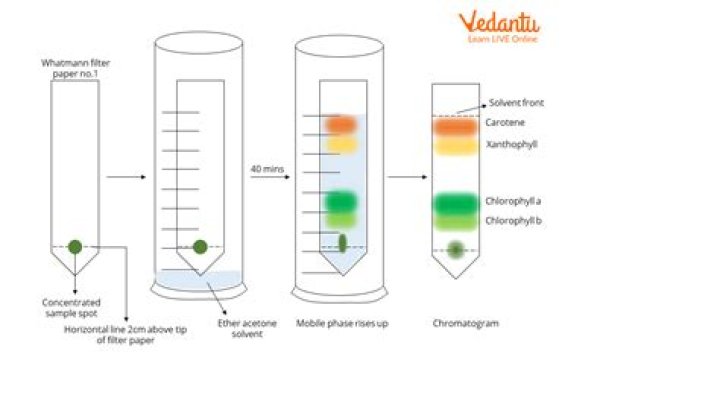
Chromatography is a method of separating mixtures by using a moving solvent on filter paper. A drop of mixture solution is spotted near one end of the paper and then dried. The end of the paper, nearest the spot, is then dipped into the solvent without submerging the spot itself.
How will you use the procedure for chromatography to identify an unknown type of colored pigment?
It is used in the sequencing of DNA and RNA. Paper chromatography is used as a qualitative analytical chemistry technique for identifying and separating colored mixtures like pigments. It is used in scientific studies to identify unknown organic and inorganic compounds from a mixture.
Related Question AnswersHow many pigments are in a leaf?
There are three types of pigments present in the leaves of plants, and their retention or production determines the colors of leaves before they fall from , molecules, beyond the simple chemical formulas that describe the numbers of atoms of different elements making up the molecule.What is an RF value?
RF value (in chromatography) The distance travelled by a given component divided by the distance travelled by the solvent front. For a given system at a known temperature, it is a characteristic of the component and can be used to identify components.Why do colors separate in paper chromatography?
As the water creeps up the paper, the colors will separate out into their components. Capillary action makes the solvent travel up the paper, where it meets and dissolves the ink. The dissolved ink (the mobile phase) slowly travels up the paper (the stationary phase) and separates out into different components.Why do pigments separate in paper chromatography?
The process of chromatography separates molecules because of the different solubilities of the molecules in a selected solvent. The solvent carries the dissolved pigments as it moves up the paper. The pigments are carried at different rates because they are not equally soluble.How do you separate pigments?
Paper chromatography is a useful technique in the separation and identification of different plant pigments. In this technique, the mixture containing the pigments to be separated is first applied as a spot or a line to the paper about 1.5 cm from the bottom edge of the paper.Which pigment has the highest RF value?
| Pigment | Rf value range | Relative position |
|---|---|---|
| Lutein | 0.22-0.28 | Below, or almost at the same level of, the highest green |
| Violaxanthin | 0.13-0.19 | Below, or almost at the same level of, the highest green |
| Neoxanthin | 0.04-0.09 | Below, or almost at the same level of, the highest green |
What is the most polar pigment?
Chlorophyll b is a more polar (water-loving) pigment than the other pigments found in spinach extracts and is therefore more strongly attracted to the polar surface of the paper than to the nonpolar solvent.Which plants and vegetables make the best dye?
Use carrots, butternut squash or giant coreopsis for a natural source of orange dye. Barberry and lilac twigs render a yellow-golden orange color. Use bloodroot to used to make a red-orange dye.How do you make a plant dye?
Mix 1 cup of salt with 16 cups of water and bring to a boil (or ½ cup of salt with 8 cups of water). Simmer your fabric in this solution for one hour prior to dyeing. (If you are making a plant/veggie based dye, mix 1 part vinegar to 4 parts water and follow the same process). When done simmering, run under cool water.Why is acetone used to extract chlorophyll pigments?
Why Is Acetone Used in Chlorophyll Extraction? Acetone breaks down chlorophyll's lipid bonds to a plant's thylakoid structure and suspends the pigment in solution. Acetone is the standard solvent for chlorophyll extraction, but ethanol, methanol, propanol, petroleum and N-dimethylformamide can also fulfill this role.How do you extract chlorophyll from plants?
Pour one cup of rubbing alcohol into the glass. Place the glass in the middle of the pot of hot water. Place the leaf in the glass, making sure that it is entirely submerged in the rubbing alcohol. After one hour, the rubbing alcohol should be green, as it now contains the chlorophyll.How do you make natural dye?
For fruit dyes, simmer the fabric in 1/4 cup salt and 4 cups water. For vegetable dyes, simmer in one cup vinegar and four cups water. Boil for one hour. Rinse the article of clothing in cold water, and then let soak in the natural dye until it reaches desired color.How do you extract beetroot color?
Bring water to a boil (med-high heat). Reduce heat. Simmer until beets are tender and there is only a few tablespoons of water left. This dyed water is your food coloring.How is chlorophyll removed from ethanol extraction?
Activated carbon (or activated charcoal) is one of the most popular options for removing chlorophyll and other unwanted non-active pigments in ethanol extracts. While activated carbon is quite effective at pigment removal, it is also very effective at removing active compounds.What is the best solvent for paper chromatography?
Readily Available Solvents for Paper Chromatography| Solvent | Polarity (arbitrary scale of 1-5) | Suitability |
|---|---|---|
| Water | 1 – Most polar | Good |
| Rubbing alcohol (ethyl type) or denatured alcohol | 2 – High polarity | Good |
| Rubbing alcohol (isopropyl type) | 3 – Medium polarity | Good |
| Vinegar | 3 – Medium polarity | Good |
What is the basic principle of chromatography?
Principle of Chromatography Chromatography is based on the principle where molecules in mixture applied onto the surface or into the solid, and fluid stationary phase (stable phase) is separating from each other while moving with the aid of a mobile phase.What are the steps in paper chromatography?
Terms in this set (7)- get ink from a real card and a questionable card.
- put samples on chromatography paper.
- hang paper in a solvent (water)
- inks will separate into colors they are made of.
- look for patterns that match.
- colors separate by solubility.
- after. retest to confirm results, decide to buy it or not.