Is AgBr covalent or ionic?
Is AgBr covalent or ionic?
Formula and structure: The chemical formula of silver bromide is AgBr and its molar mass is 187.77 g/mol. It is an inorganic compound made up of the silver metal (Ag) and the bromine atom (Br), held together through a polar covalent bond which has a strong ionic character.
How do you determine if a bond is ionic or covalent?
Compounds containing two elements (so called binary compounds) can either have ionic or covalent bonding.
- If a compound is made from a metal and a non-metal, its bonding will be ionic.
- If a compound is made from two non-metals, its bonding will be covalent.
How do you determine if a compound is ionic or covalent electronegativity?
If the difference of the electronegativity between the two elements is greater than 1.7 then the bond is ionic. The difference with a polar covalent bond is 0.5 to 1.7 and a nonpolar covalent bond is from 0 to 0.4.
Is RbBr ionic?
Ionic compounds Some examples are NaCl (table salt), KCl, CsF, and RbBr. Each is composed of a positive ion and a negative ion.
Why is silver iodide covalent?
In AgI , due to large size of Iodine than chlorine(all other factors are same), it is highly polarizable and hence has higher covalent character .
Is AgCl ionic or covalent?
Hence AgCl is ionic in nature and referred as an ionic compound. Note:The bonds formed between the atoms which have electronegativity difference greater 1.7 will be ionic and polar in nature. The sharing of electrons between the atoms will result in a covalent bond.
How do you calculate covalent bonds?
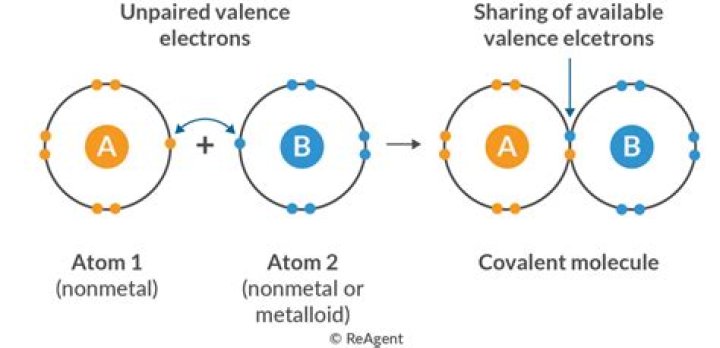
A covalent bond is formed between two atoms by sharing electrons. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet. Hydrogen is an exception to the octet rule. H forms only one bond because it needs only two electrons.
How do you determine bond type?
Bond type is predicated on the difference in electronegativity of the two elements involved in the bond. The degree to which a given bond is ionic or covalent is determined by calculating the difference in electronegativity between the two atoms involved in the bond.
Is HBr ionic or covalent?
Therefore the HBr gas has a covalent bond polarized and the hydrogen atom has a slight positive charge and Br has a slight negative charge. In fact, the molecular orbital is attracted toward the bromine.
Is nacl covalent?
For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals….Table 2.11.
| Property | Ionic | Covalent |
|---|---|---|
| Melting temperature | High | Low |
Is sio2 an ionic bond?
Silicon dioxide has a giant covalent structure. Part of this structure is shown in the diagram – oxygen atoms are shown as red, silicon atoms shown as brown: Each silicon atom is covalently bonded to four oxygen atoms. Each oxygen atom is covalently bonded to two silicon atoms.
Is silver bromide ionic or covalent?
Formula and structure: The chemical formula of silver bromide is AgBr and its molar mass is 187.77 g/mol. It is an inorganic compound made up of the silver metal (Ag) and the bromine atom (Br), held together through a polar covalent bond which has a strong ionic character.
Is silver chloride ionic or covalent bond?
Answer: silver chloride ( AgCl ) is ionic bond. What is chemical bond, ionic bond, covalent bond? Chemical bond. A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds.
How do you make silver bromide naturally?
Occurrence: Silver bromide occurs naturally as the mineral bromargyrite in considerable amounts. However, it is typically obtained in large amounts through chemical production. Preparation: Silver bromide is prepared industrially by the reaction between aqueous solutions of silver nitrate and potassium bromide.
What is the GHS classification for silver bromide?
More… Silver bromide, Premion? H410 (100%): Very toxic to aquatic life with long lasting effects [ Warning Hazardous to the aquatic environment, long-term hazard] (The corresponding statement to each P-code can be found at the GHS Classification page.)