Biphenyl was partially soluble in methanol which is intermediately polar whilst biphenyl is nonpolar, allowing it to dissolve a little. However, biphenyl is less polar than benzophenone. Since methyl alcohol is somewhat polar, the more polar benzophenone is more soluble in methyl alcohol than biphenyl..
Also asked, is benzophenone soluble in methanol?
Benzophenone, which is largely nonpolar, but possesses a polar carbonyl group, was found to be partially soluble in methyl alcohol and hexane but insoluble in water.
Furthermore, is methanol polar or nonpolar? Methanol is a polar molecule: The alcohol (-OH) group dominates the molecule making it definitely polar. The electrostatic potential clearly shows that the oxygen is partially negative whereas the carbon and hydrogens are partially positive.
One may also ask, is methanol soluble in hexane?
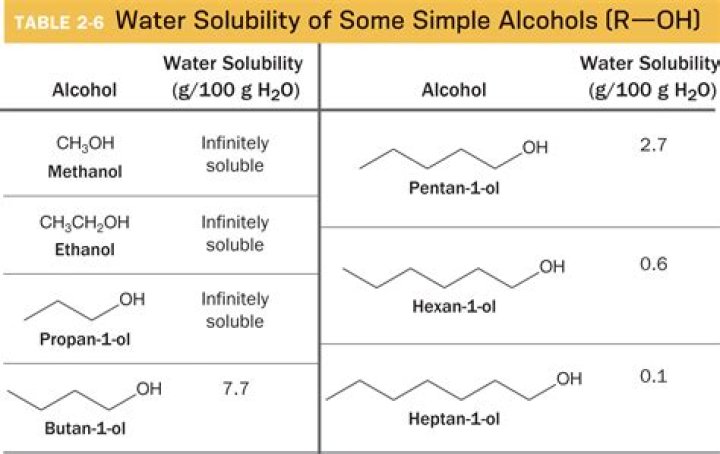
1-methanol has only one carbon and the –OH group makes a polar molecule. Since water is polar, and like dissolves in like, 1-methanol is soluble in water. 1-octanol and 1-butanol in hexane are both soluble because hexane is a non-polar molecule.
Does biphenyl dissolve in water?
Biphenyl is insoluble in water, but soluble in typical organic solvents. The biphenyl molecule consists of two connected phenyl rings.
Related Question Answers
Is HCl polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?Why does benzophenone dissolve in methanol?
The polarity of methyl alcohol lies somewhere between polar and non-polar. Benzophenone contains a polar carbonyl group that forms the hydrogen bonding with the OH group of methyl alcohol. So due to this intermolecular hydrogen bonding benzophenone is soluble in methyl alcohol.Why is methanol not soluble in hexane?
Because hexane is non-polar (dipole moment is 0) and methanol is pretty polar (dipole moment 5.5 E-30Cm). Nonpolar solvents like hexane are not mixing well with polar solvents like methanol.Is vanillin polar or nonpolar?
The most common use for vanillin (vanilla extract) is as a flavoring agent for food and as an aromatic agent in some perfumes. It can also serve as a chemical intermediate for the production of pharmaneuticals. The bonds between C - H, C -C are nonpolar, but the O - C and the single O - H bond is polar.Is acetone polar or nonpolar?
Water is a polar molecule, while carbon tetrachloride is a nonpolar molecule. If we use the 'like dissolves like' concept, then acetone is both polar and nonpolar. This dual character of acetone is due to its ability to make hydrogen bonds with water and the presence of two methyl groups.Why is ethyl 4 Aminobenzoate soluble in HCl?
However, when 6.0 M HCl (strong acid) is added to the basic solvent, benzoic acid is no longer soluble. Conversely, Ethyl 4-aminobenzoate (base) is soluble when mixed with 1.0 HCl. However, when 6.0 M NaOH is added to the 1.0 M HCl mixture, Ethyl 4-aminobenzoate is no longer soluble and forms a precipitate.Is benzoin or benzil more polar?
Since benzil is less polar than benzoin, a polar solvent is used as solvent for the recrystallization.Is 1 octanol polar or nonpolar?
The molecule has many more nonpolar bonds than polar bonds, so overall the molecule is considered as nonpolar (or only very slightly polar). For 1-octanol ε = 10 (nonpolar). The biological properties of lipids can be understood due to their overall nonpolar nature.Why Methanol is soluble in water?
Originally Answered: why methanol is soluble in water? Because of the presence of only one hydrophobic carbon, it can form hydrogen bonds with water molecules and hence it is soluble in water.What is soluble in hexane?
For example, ionic compounds, which are very polar, are often soluble in the polar solvent water. Nonpolar substances are likely to dissolve in nonpolar solvents. For example, nonpolar molecular substances are likely to dissolve in hexane, a common nonpolar solvent. For example, ionic compounds are insoluble in hexane.Is pentane soluble in methanol?
The pentanes are molecular, and certainly they are non-polar, and hence addition of n-pentane to water gives rise to two immiscible layers, with the LESS dense pentane floating on top of the funnel. And contrast this with the solubility of ethanol and methanol in water.Is methanol soluble in water?
The water molecule is polar and hydrogen bonding exists between the molecules of water. As far as intermolecular bonding forces are concerned hydrogen bonding is relatively strong. Methanol is soluble in water. Like water, methanol has relatively strong hydrogen bonding acting between its molecules.Is ethanol more polar than methanol?
My review book (Princeton review) says that ethanol has stronger intermolecular forces than methanol because it has a large molecular mass and is therefore more polarizable and more soluble in water than methanol. Therefore ethanol is less polar.Is ethanol soluble in methanol?
Ethanol has a 2 carbon chain and a OH group. As water is polar it attracts OH group. Solubility of alcohols is therefore determined by the stronger of the two forces. Because of the strength of the attraction of the OH group, first three alcohols (methanol, ethanol and propanol) are completely miscible.Is butanol polar or nonpolar?
n-Butanol is soluble in water. It is a polar molecule unlike what others have suggested. It's not as soluble as shorter chain alcohols such as methanol. The polar part is the -OH group whilst the non polar part is the greasy hydrocarbon chain.In which solvent would lipids be most soluble?
Lipids are all insoluble in polar solvents like water but highly soluble in the non-polar or weakly polar organic solvents, including ether, chloroform, benzene, and acetone.Is oil soluble in ethanol?
of alcohol. Mowrah, safflower, peanut, and cottonseed oils are soluble in absolute alcohol at 70°C. In 95% ethanol they are miscible between 90° and 100°C. Addition of a good solvent, like n-hexane, increases the solubility of oils, and the solubility temperatures are lowered.Is water non polar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5.Is methanol a hydrogen bond?
Methanol with hydrogen bonding: Methanol is a polar molecule, with the oxygen (red) being the negative area and the hydrogen (white) being the more positive area. Opposite charges attract. Some combinations which are not hydrogen bonds include: hydrogen to another hydrogen or hydrogen to a carbon.