Is ethyl acetate more polar than acetone?
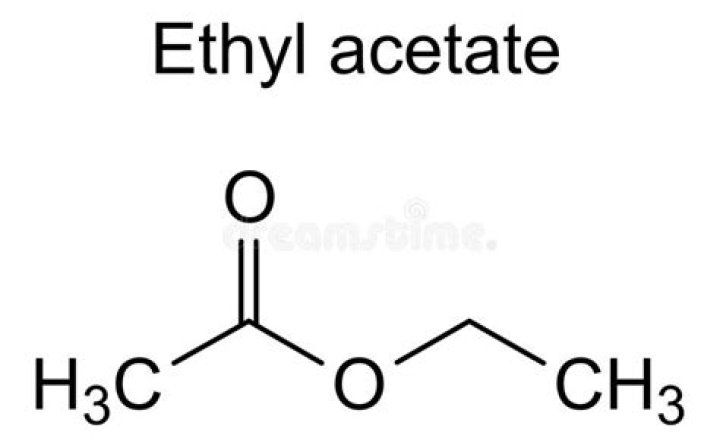
.
In this way, is acetone more polar than ethanol?
It is true that acetone is less polar than ethanol. But the dipole moment of acetone is higher than ethanol.
Likewise, is acetone polar or nonpolar? Water is a polar molecule, while carbon tetrachloride is a nonpolar molecule. If we use the 'like dissolves like' concept, then acetone is both polar and nonpolar. This dual character of acetone is due to its ability to make hydrogen bonds with water and the presence of two methyl groups.
Subsequently, question is, is ethyl acetate the same as acetone?
Acetone is a clear, harsh-smelling and highly flammable liquid. It's a solvent, capable of disintegrating even plastic. The key active ingredient in non-acetone removers is usually ethyl acetate. Made from ethanol and acetic acid, ethyl acetate is colorless and also flammable.
Is ethyl acetate more polar than acetic acid?
Solvents and Polarity. Workup for Polar and Water-Soluble Solvents.
Solvents and Polarity.
| Solvent | Relative Polarity |
|---|---|
| acetic acid | 0.648 |
| ethanol | 0.654 |
| methanol | 0.762 |
| ethylene glycol | 0.79 |
What is the most polar solvent?
Polar protic solvents Examples include water, most alcohols, formic acid, hydrogen fluoride, and ammonia. Polar protic solvents are favorable for SN1 reactions, while polar aprotic solvents are favorable for SN2 reactions.Is water more polar than ethanol?
Because water molecules are polar, they connect through hydrogen bonds. Alcohol is much less polar than water. Because it's non-polar, the molecules don't form hydrogen bonds.Is acetone more polar than hexane?
Answer: Acetone is a more polar solvent than is hexanes. If it were used to elute the same three compounds, each of the compounds would travel faster because the more polar eluting solvent is more proficient at eluting the compounds from the polar adsorbent.Is ethanol polar or nonpolar?
Ethanol is a very polar molecule due to its hydroxyl (OH) group, with the high electronegativity of oxygen allowing hydrogen bonding to take place with other molecules. Ethanol therefore attracts polar and ionic molecules. The ethyl (C2H5) group in ethanol is non-polar. Ethanol therefore attracts non-polar molecules.Is ethanol more polar than methanol?
My review book (Princeton review) says that ethanol has stronger intermolecular forces than methanol because it has a large molecular mass and is therefore more polarizable and more soluble in water than methanol. Therefore ethanol is less polar.Why acetonitrile is more polar than methanol?
Methanol is a polar-protic solvent, whereas acetonitrile is a polar-aprotic solvent and possesses a stronger dipole moment. This means that the organic modifier used in the mobile phase can have a powerful effect on chromatographic selectivity.Is benzene polar or nonpolar?
We could say that benzene is nonpolar because it is a hydrocarbon that contains only C-C and C-H bonds, and hydrocarbons are nonpolar. But C is slightly more electronegative than H (by 0.35 units), so a C-H bond is very slightly polar and has a small dipole moment.How dangerous is ethyl acetate?
Ethyl acetate is highly flammable, as well as toxic when ingestion or inhaled, and this chemical can be seriously damaging to internal organs in the case of repeated or prolonged exposure. Ethyl acetate can also cause irritation when it comes into contact with the eyes or skin.Why is ethyl acetate a good solvent?
Ethyl Acetate Uses As a high purity solvent, it is used in cleaning electric circuit boards and as a nail polish remover. At a lower purity, it can be used as in printing, pharmaceuticals, perfumes, food, decaffeination of tea/coffee and a carrier solvent for herbicides.What does acetone do to your body?
Breathing moderate to high amounts of acetone for a short amount of time can irritate your nose, throat, lungs and eyes. It can also cause headaches, dizziness, confusion, a faster pulse, nausea, vomiting, effects on the blood, passing out and possible coma, and a shorter menstrual cycle in women.What products contain acetone?
Common Products Containing Acetone- Nail polish and nail polish removers.
- Hair colors.
- Hair tonics and conditioners.
- Manicuring preparations and other toiletries.
- Sun tan and other lotions applied on the skin.
- Baby wipes.
- Pre-moistened towels.
- Art material including clay, finger paints, and tempera colors.



