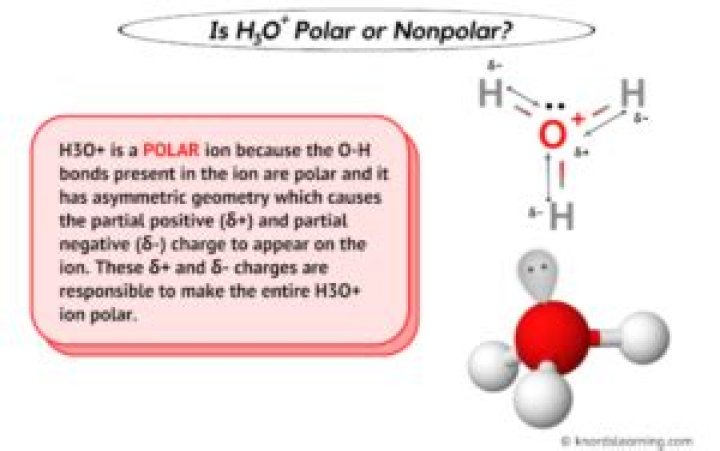
The overall molecule is Polar because the shape of the molecule is Trigonal Pyramidal, which means it has the lone pair electrons. Becuase of the lone pair the pulling is unequal. H3O+ has 3 polar bonds. So H3O+ has 3 polar bonds, and the overall molecule is polar too..
People also ask, what type of bond is h3o+?
Lewis Dot of Hydronium H3O+ Hydronium is the positive ion present in Arrhenius acid solutions. It is formed from a hydrogen ion and water bonding. Hydronium contains 2 polar covalent bonds and 1 coordinate covalent bond.
Additionally, what is the shape of h3o positive? The hydronium ion has a trigonal pyramidal geometry and is composed of three hydrogen atoms and one oxygen atom. There is a lone pair of electrons on the oxygen giving it this shape. The bond angle between the atoms is 113 degrees.
Thereof, is co2 polar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.
Is h3o an acid or base?
The H3O+ is the conjugate acid of H2O. So H3O+ is used as a shorthand for a proton in aqueous solution. In a non-aqueous solution the proton would form a different structure. Shows that H2O is made up of equal parts H+ and OH- ions and is amphoteric (can be an acid or a base) having a deprotonated form (OH-).
Related Question Answers
Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?Can you drink h3o?
There is no such thing as uncharged H3O, but if you really mean H3O+, not only can you drink it, you do so every day. The hydronium ion itself is a very strong acid, the strongest that can exist in an aqueous solution.Why is ClF polar?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). For these molecules, the applicable intermolecular interactions are dipole and induced dipole interactions.Is h2o ionic or covalent?
H2O, more commonly known as water, is a covalent compound. This type of compound is the result of atoms, usually from nonmetal elements, sharing electrons. Water has a special type of covalent bond called a polar covalent bond.Is HBr polar or nonpolar?
Because HBr has two opposite sides of polarity, it is a POLAR MOLECULE. Polar molecules stick to their neighbors and tend to have higher boiling points. Bonds between two identical atoms have a zero electronegativity difference. They are 100% nonpolar covalent.How do I calculate bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order: - Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
What is a dipolar bond?
A dipolar bond, more commonly known as a dative covalent bond or coordinate bond is a kind of 2-center, 2-electron covalent bond in which the two electrons derive from the same atom.What is the shape of co2?
linear
Is ozone a polar molecule?
Ozone is a polar molecule with a dipole moment of 0.53 D.What kind of bond is co2?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, which is shown here as two lines and referred to as a double bond. When molecules are symmetrical, however, the atoms pull equally on the electrons and the charge distribution is uniform.Is water Polar?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.What is the electronegativity of co2?
The carbon-oxygen double bonds in the linear CO2 molecule are polar (electronegativities: C = 2.5, O = 3.5). The electrons in each of the double bonds are drawn toward the oxygens, so both oxygen atoms have a partial negative charge.Why is CH3Cl polar?
Since the H is between B and C in terms on electronegativity values, their difference in electronegativity values is so small, the C-H bond is considered nonpolar; thus, no dipole arrow is drawn for the C-H bonds. Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.Is oxygen polar or nonpolar?
However, in Oxygen molecule two oxygen atoms are bonded. So, there is no question of electronegativity difference, electron shift and partial charge creation. So, the bond between 2 Oxygen atom is non-polar . Since, it is the only bonding in oxygen molecule Oxygen molecule is non-polar.Are double bonds polar or nonpolar?
This is a linear molecule, containing two polar carbon-oxygen double bonds. However, since the polar bonds are pointing exactly 180° away from each other, the bond polarities cancel out, and the molecule is nonpolar.Is co2 a dipole molecule?
A molecule like CO2 may be composed of two dipoles, but it has no dipole moment. This is because the charge is equally distributed amongst the entire molecule. When molecules have an even charge distribution and no dipole moment, then they are nonpolar molecules.What is the bond angle of h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.Is BrF3 planar?
(a) CO, (c) BrF3, BBr3, like BF3, is trigonal planar and the B-Br bond dipoles cancel out. The same is true for trigonal bipyramidal PF5. BrF3 is a distorted T-shape, and the bond dipoles don't cancel out.What is the molecular geometry of h30+?
In H3O+, the central atom ( oxygen ) has three bond pairs and one lone pair and hence, it is sp3 hybridized. You must be knowing that ideal geometry for a sp3 hybridized central atom molecule is Tetrahedral.