Is Li2O an ionic compound?
.
Then, what type of bond is Li2O?
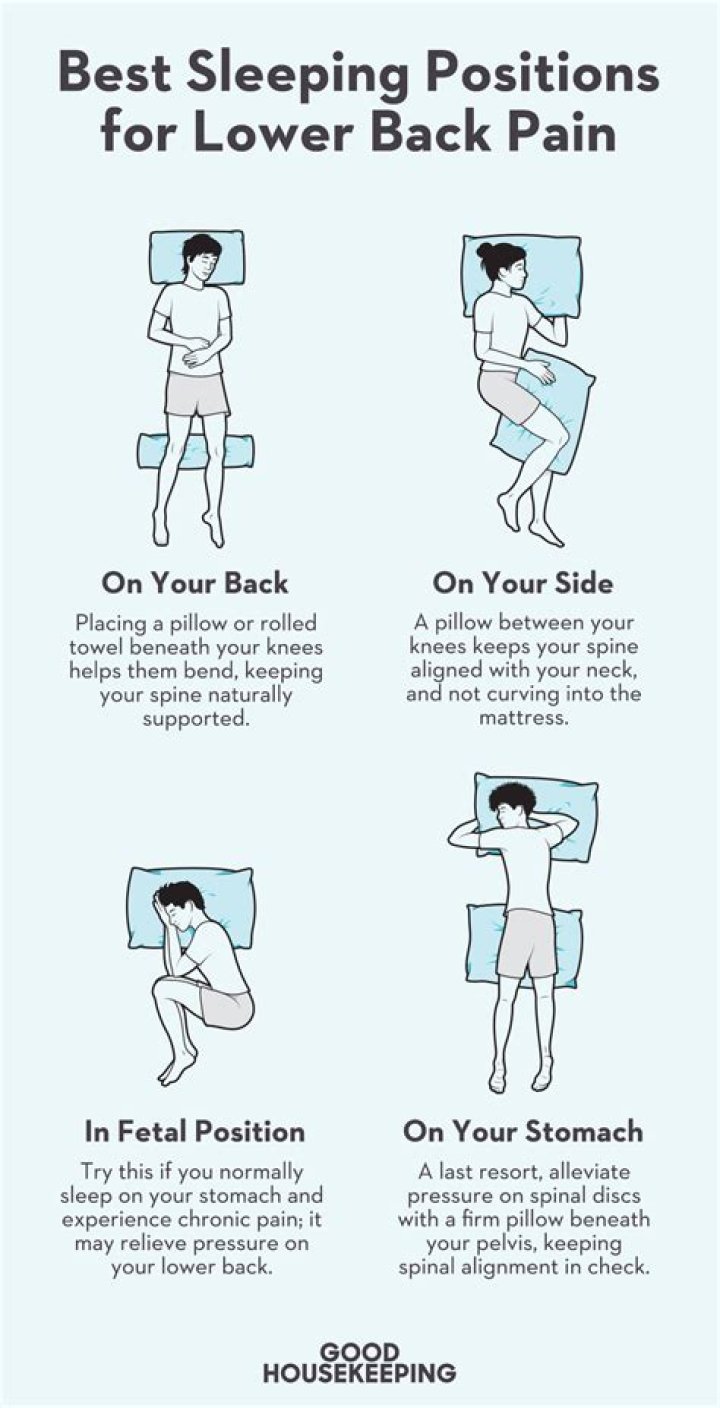
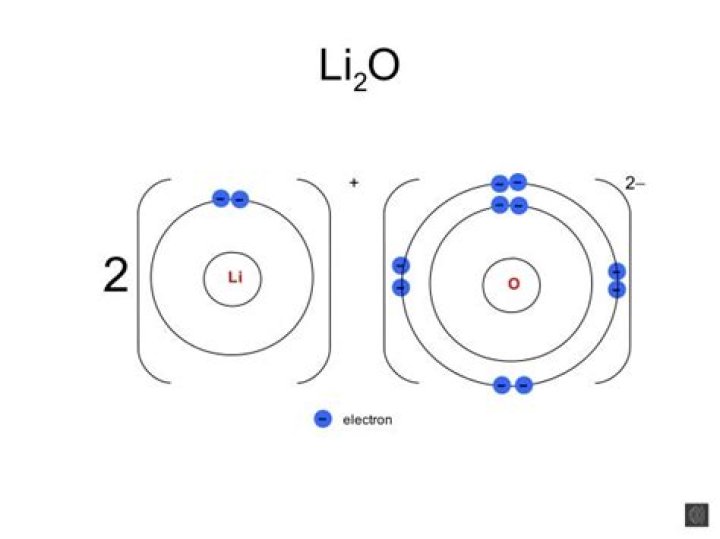
This is how the ionic bond forms in Lithium Oxide (Li2O).
Secondly, is pcl3 covalent or ionic? In CCl4, atoms chlorine and carbon share electron pairs to form a covalent bond. Ionic bonds are formed by electrostatic interactions between an electropositive and electronegative atom.
Besides, is Lithium oxide an ionic compound?
Lithium oxide is an ionic compound formed between a metal (Li) and a non- metal (O) by the complete transfer of electrons from Li to O to give Li+ cations and O2– anions. These ions are held in place in the crystal lattice by strong electrostatic attractions between the positively and negatively charged ions.
Is caf2 ionic or covalent?
Calcium fluoride (CaF2) is an insoluble ionic compound composed of Ca2+ and F− ions. It occurs naturally as the mineral “Fluorite” (also called fluorspar) and as “Blue-John”. This salt is the source of most of the world's fluorine.
Related Question AnswersIs HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.Is AlCl3 ionic or covalent?
Fluorine (being very electronegative) will "steal" a lot of electron density from aluminium making the bonds more ionic in nature than covalent. But AlCl3 is covalent due to the low difference in electronegativity. Hence proved! Anhydrous AlCl3 is covalent in nature.Why is Li2O linear?
The shape of Li2O will be linear. In Valence Shell Electron Pair Repulsion (VSEPR) theory, pairs of electrons that surround the central atom of a molecule or ion are arranged as far apart as possible to minimise electron-electron repulsion.Is NaF a polar covalent bond?
Bonds with partially ionic and partially covalent character are called polar covalent bonds. Formation of Sodium FlourideThe attraction of oppositely charged atoms and the transfer of electrons leads to the formation of an ionic compound. In this case, NaF.What are the properties of ionic compounds?
Properties Shared by Ionic Compounds- They form crystals.
- They have high melting points and high boiling points.
- They have higher enthalpies of fusion and vaporization than molecular compounds.
- They're hard and brittle.
- They conduct electricity when they are dissolved in water.
- They're good insulators.
Is Li2O a covalent compound?
For example, the systematic name of H2O is dihydrogen monoxide, while the systematic name of Li2O is lithium oxide – one has covalent bonds and uses the prefix di- and the other has ionic bonds and does not use prefixes.Is Na2O a covalent bond?
Sodium oxide (Na2O) is an ionic compound. Its constituent atoms are, respectively, a metal and a non-metal. A transfer of valence electrons will occur when a bond forms, thus making the compound ionic in bonding nature.What holds ions together?
Ionic Bonding. An ionic bond is held together by the electrostatic attraction between ions that are near one another. Electrostatic attraction is the attraction between atoms that have opposite charge and holds the atoms together in ionic bonds.How do you name ionic compounds?
Method 1 Naming Basic Ionic Compounds- Jot down the formula of the ionic compound. Let's say the ionic compound you're working with is NaCl.
- Write the name of the metal.
- Add the name of the non-metal with an –ide ending.
- Combine the cation and anion names.
- Practice naming more simple ionic compounds.