Is potassium fluoride an acid or base?
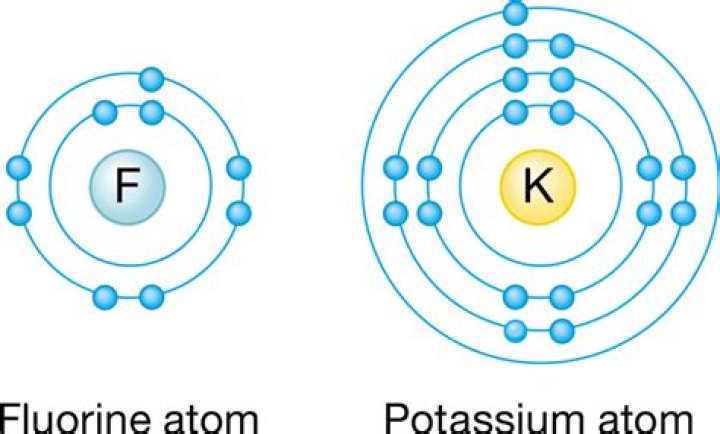
Is potassium fluoride an acid or base?
HF is a WEAK acid in aqueous solution……. i.e. Ka=10−3.17=6.76×10−4 . If we describe something as a weak acid, we might also describe its conjugate base as reasonably strong in that it competes for the proton effectively.
How do you know if its acid or base?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).
What kind of substance is potassium fluoride?
Potassium fluoride, represented by the chemical formula KF, is an inorganic compound comprising an alkali metal potassium and monoatomic anion fluoride. It exists in its solid state or aqueous solution form, with the mineral carobbiite being the naturally occurring KF.
Is Hydrosulfuric acid a strong acid?
Hydrosulfuric acid is classified as a weak acid. Its chemical symbol is H2S H 2 S .
Is potassium fluoride a weak base?
KF is a salt that comes from a strong base, KOH, and a weak acid, HF. This is to be expected as KF can be thought of being formed in the reaction of a weak acid (HF) with a strong base (KOH). Acid with values less than one are considered weak. KF is a salt that comes from a strong base, KOH, and a weak acid, HF.
Is potassium fluoride an ionic compound?
Potassium fluoride is an ionic compound formed from the transfer of 1 valence electron from potassium to fluorine. This ionic bonding is possible because potassium is metal while fluorine is non-metal. Since cation potassium is K+ while the anion fluorine is F− , the chemical formula of potassium fluoride is KF .
How do you know if an acid or base is pH?
A solution’s pH will be a number between 0 and 14. A solution with a pH of 7 is classified as neutral. If the pH is lower than 7, the solution is acidic. When pH is higher than 7, the solution is basic.
How do you know if its a weak acid or base?
Key Takeaways
- Strong acids and bases are 100% ionized in aqueous solution.
- Weak acids and bases are less than 100% ionized in aqueous solution.
- Salts of weak acids or bases can affect the acidity or basicity of their aqueous solutions.
Is potassium fluoride soluble or insoluble?
Potassium fluoride
| Names | |
|---|---|
| Solubility in water | anhydrous: 92 g/100 mL (18 °C) 102 g/100 mL (25 °C) dihydrate: 349.3 g/100 mL (18 °C) |
| Solubility | soluble in HF insoluble in alcohol |
| Magnetic susceptibility (χ) | −23.6·10−6 cm3/mol |
| Structure |
What is potassium fluoride in?
Potassium Fluoride is a white, crystalline (sand-like) powder with a sharp, salty taste. It is used in etching glass, as a preservative, and as an insecticide.
Is potassium hydroxide a strong base?
Strong bases A strong base is something like sodium hydroxide or potassium hydroxide which is fully ionic. You can think of the compound as being 100% split up into metal ions and hydroxide ions in solution.
Is hydroiodic acid strong or weak?
Hydroiodic acid (or hydriodic acid) is an aqueous solution of hydrogen iodide (HI). It is a strong acid, one that is ionized completely in an aqueous solution.