Is rutherfordium a solid at room temperature?
.
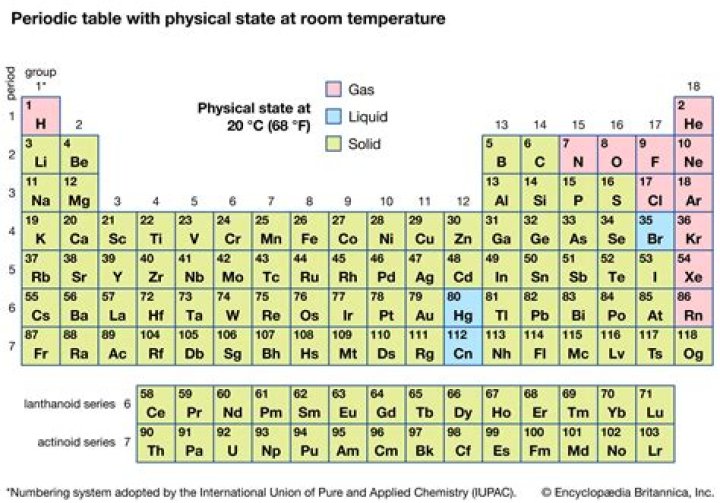
Hereof, what state of matter is rutherfordium at room temperature?
solid
Additionally, what is rutherfordium natural state? Rutherfordium is a synthetic chemical element with the symbol Rf and atomic number 104, named after New Zealand physicist Ernest Rutherford. As a synthetic element, it is not found in nature and can only be created in a laboratory. The chemical properties of rutherfordium are characterized only partly.
Likewise, people ask, is rutherfordium radioactive?
Rutherfordium. Rutherfordium is a highly radioactive chemical element. His most stable isotope Rf 265 has an half like of approximately 13 hours. Rutherforium is not found free in the environment, since it is a synthetic element.
Where is rutherfordium found?
Rutherfordium was discovered by Workers at the Nuclear Institute at Dubna, and the University of California, Berkeley, USA. in 1964 at USSR, United States. Origin of name: the origin of the Amercian Chemical Society preferred name is Lord "Rutherford", the physicist and chemist from New Zealand..
Related Question AnswersHow long does Bohrium last?
The most stable is 270Bh, which has a half-life of about one minute. The atomic weight for manmade transuranium elements is based on the longest-lived isotope in the periodic table. These atomic weights should be considered provisional since a new isotope with a longer half-life could be produced in the future.What is made out of rutherfordium?
Source: Rutherfordium is a synthetic radioactive metal, created by nuclear bombardment, and has only been produced in tiny amounts. Rutherfordium can be made by bombarding plutonium-242 with accelerated neon ions or by bombarding californium-249 with accelerated carbon ions.Where is Dubnium found?
Named after Dubna, the area near Moscow where Soviet scientists discovered rutherfordium and dubnium. Dubnium is a highly radioactive metal. It has never been found naturally and only a small number of atoms have been produced in laboratories.What is the classification of rutherfordium?
Rutherfordium is a chemical element with symbol Rf and atomic number 104. Classified as a transition metal, Rutherfordium is a solid at room temperature.Who discovered rutherfordium?
Albert Ghiorso Joint Institute for Nuclear ResearchAre there man made elements?
A synthetic element is one of 24 chemical elements that do not occur naturally on Earth: they have been created by human manipulation of fundamental particles in a nuclear reactor or particle accelerator, or explosion of an atomic bomb; thus, they are called "synthetic", "artificial", or "man-made".Is rutherfordium man made?
Rutherfordium is a man-made element first claimed to have been observed in 1964 in a particle accelerator in Dubna in Russia by fusing ions of plutonium (atomic number 94) and neon (atomic number 10). This was later disputed by American scientists who made it in 1969 by a completely different method.What is vanadium used for?
Ferrovanadium and vanadium-steel alloys are used to make such things as axles, crankshafts and gears for cars, parts of jet engines, springs and cutting tools. Vanadium pentoxide (V2O5) is perhaps vanadium's most useful compound. It is used as a mordant, a material which permanently fixes dyes to fabrics.Is Dubnium radioactive?
Source: Dubnium is a synthetic radioactive metal, created by nuclear bombardment, and has only been produced in tiny amounts. The most stable isotope is 268Db, with a half-life of 32 hours.Why is rutherfordium important?
Because it has been around for longer and its isotopes are better known, more is known about the chemistry of rutherfordium than of any succeeding element. Working with rutherfordium requires specialist methods and knowledge, as it involves working with tiny quantities of very short-lived, radioactive atoms.What was seaborgium named after?
Seaborgium is a synthetic chemical element with the symbol Sg and atomic number 106. It is named after the American nuclear chemist Glenn T. Seaborg. As a synthetic element, it can be created in a laboratory but is not found in nature.What does Oganesson look like?
Oganesson is a radioactive, artificially produced element about which little is known. It is expected to be a gas and is classified as a non-metal. It is a member of the noble gas group. The IUPAC also approved names for elements 113 (nihonium, with atomic symbol Nh), 115 (moscovium, Mc) and 117 (tennessine, Ts).What research is rutherfordium used for?
Uses of Rutherfordium Because rutherfordium is made within the lab, there are not very many uses for this element commercially. On the other hand, rutherfordium has been used within the laboratory setting to conduct research. Most elements that are highly radioactive are used for nuclear power and medicinal purposes.What is the electron configuration of rutherfordium?
Rn 5f14 6d2 7s2How did Moscovium get its name?
According to IUPAC recommendations, the discoverer(s) of a new element has the right to suggest a name. A suggested name was langevinium, after Paul Langevin. Later, the Dubna team mentioned the name moscovium several times as one among many possibilities, referring to the Moscow Oblast where Dubna is located.What family is rutherfordium in?
| Name | Rutherfordium |
|---|---|
| Density | Unknown |
| Normal Phase | Synthetic |
| Family | Transition Metals |
| Period | 7 |