What are examples of acids and alkalis?
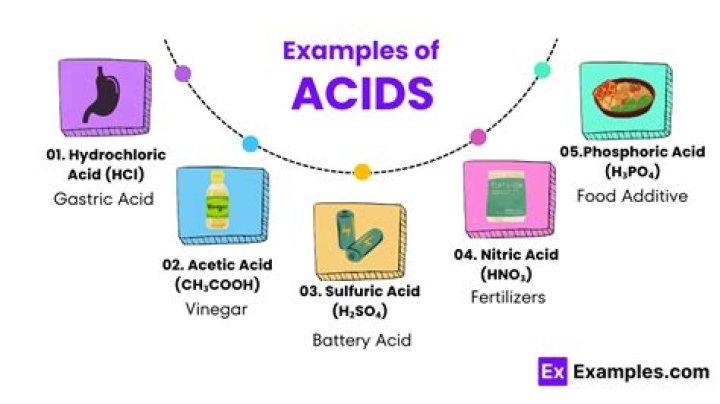
Common lab acids include:
- Hydrochloric acid.
- Sulfuric acid.
- Nitric acid.
.
Similarly, what are the examples of alkalis?
An alkali is a basic hydroxide or ionic salt of an alkali metal or alkaline earth metal element, which is soluble in water. Only few alkalis are known, examples, sodium hydroxide (NaOH), potassium hydroxide (KOH), calcium hydroxide (Ca(OH)2), calcium carbonate (CaCO3), and magnesium hydroxide (Mg(OH)2.
Likewise, what acids and alkalis are used at home? Table 7: Alkalis and acids frequently found in household products
| Alkalis | Products |
|---|---|
| Acids | Products |
| Sulfuric acid Hydrochloric acid Sodium bisulfate Sodium hypochlorite Acetic Acid Nitric acid | Drain cleaner Toilet bowl cleaner House bleach (low concentration) Descalers |
Furthermore, what are acid and alkalis?
Acids and alkalis both contain ions. Acids contain lots of hydrogen ions, which have the symbol H+. Alkalis contain lots of hydroxide ions, symbol OH-. Water is neutral because the number of hydrogen ions is equal to the number of hydroxide ions.
How do you identify acids and alkalis?
Acids, Alkalis and Neutral Substances
- They are liquids.
- They are solutions of compounds in water.
- If concentrated they can be corrosive.
- Acids taste sour (for example, vinegar).
- Turn blue litmus paper red - this is an easy test for an acid!
- Usually react with metals to form salts.
- Acids contain hydrogen ions.
Is toothpaste an alkali?
Toothpaste is alkaline. In order to neutralise this acidic effect, we brush out teeth with toothpaste. Since it is used for neutralising acids, a toothpaste can never be acidic, it is basic or alkaline.What is called alkali?
In chemistry, an alkali (/ˈælk?la?/; from Arabic: al-qaly "ashes of the saltwort") is a basic, ionic salt of an alkali metal or alkaline earth metal chemical element. An alkali also can be defined as a base that dissolves in water. A solution of a soluble base has a pH greater than 7.0.Are alkalis dangerous?
Just like concentrated acids, concentrated alkalis are corrosive. They can attack metals and destroy skin if spilled, so their containers are labelled with a warning symbol. Concentrated alkalis are just as dangerous as concentrated acids, sometimes more dangerous, but many people do not realise this.What are alkalis used for?
Uses of common Alkalis Sodium hydroxide is used to make paper, detergents and soap. Potassium hydroxide is used in farming to make acidic soil more alkaline so that plants will grow better in it, and is also used as the electrolyte in alkaline, Ni-Cd, and Ni-MH batteries.What is alkali made of?
Alkali manufacture is the process by which an alkali is made. Typical alkalis, produced commercially, include sodium hydroxide, sodium carbonate, potassium hydroxide and potassium carbonate.What are 3 weak bases?
Weak Acids & Bases| Common Weak Acids | Common Weak Bases | |
|---|---|---|
| Acetic | CH3COOH | trimethyl ammonia |
| Trichloroacetic | CCl3COOH | pyridine |
| Hydrofluoric | HF | ammonium hydroxide |
| Hydrocyanic | HCN | water |
How many alkalis are there?
So, the eight commonly known alkalis are LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2 and Ba(OH)2. Is copper(II) sulfate soluble in water?What is the base?
In chemistry, a base is a chemical species that donates electrons, accepts protons, or releases hydroxide (OH-) ions in aqueous solution. Types of bases include Arrhenius base, Bronsted-Lowry base, and Lewis base.What are five properties of acids?
Acids- Aqueous solutions of acids are electrolytes, meaning that they conduct electrical current.
- Acids have a sour taste.
- Acids change the color of certain acid-base indicates.
- Acids react with active metals to yield hydrogen gas.
- Acids react with bases to produce a salt compound and water.
How do acids feel?
Acids taste sour while bases taste bitter. An acid reacts with metals to produce bubbles of hydrogen gas while a base feels slimy to the touch.What are acids and alkalis used for?
We use acids and alkalis in our daily lives for things like cleaning, cooking and you even eat and drink some substances that are acidic or alkaline. Common lab acids include: Hydrochloric acid. Sulfuric acid.Which is more harmful acid or alkali?
Substances with pH values less than seven are acids, while numbers higher than seven are alkaline. The higher or lower the number, the more acidic or basic a substance is and the more damage it can cause. Alkali burns are the most dangerous.Is vinegar an acid?
Summary Vinegar is mildly acidic with a pH of 2–3. Apple cider vinegar is slightly more alkaline than pure vinegar because it contains more alkaline nutrients. However, it's still acidic.Is table salt neutral?
The prototype “salt,” of course, is sodium chloride, or table salt. Sodium chloride, which is obtained by neutralization of hydrochloric acid and sodium hydroxide, is a neutral salt. Neutralization of any strong acid with a strong base always gives a neutral salt.What is acids and alkalis year 7?
A pH of 7 is neutral (like for example water); a pH of 1 or 2 is a strong acid (for example sulfuric acid); weak acids like vinegar have a pH of 3 to 6. Weak alkalis (like soap) have pH values of 8 to 10 and a strong alkali like sodium hydroxide will have a pH of 13 or 14.What liquids are neutral?
Water and human blood are great examples of neutral solutions. Acids mixed with bases can be neutralized and given a pH of 7. Water can break down to form (H+) and (OH-) ions. When these ions are equal to each other, the value is 1 *10^-7, or neutral.What substances are acids?
An acid is a substance that donates hydrogen ions. Because of this, when an acid is dissolved in water, the balance between hydrogen ions and hydroxide ions is shifted. Now there are more hydrogen ions than hydroxide ions in the solution. This kind of solution is acidic.What are some strong alkalis?
Alkalis are very corrosive in nature and penetrate deeply. Examples of strong alkalis (lyes) include barium, sodium, ammonium, calcium, lithium, and potassium hydroxides.What alkalis are in the home?
Examples of common alkalis:- caustic soda (sodium hydroxide): used to clean ovens and drains.
- washing soda (sodium carbonate)
- ammonia solution (often used in household cleaners)
- garden lime (calcium oxide): used to neutralise acid in the soil.
- indigestion powder (often magnesium hydroxide)