What are examples of buffer systems?
What are examples of buffer systems?
A buffer system can be made of a weak acid and its salt or a weak base and its salt. A classic example of a weak acid based buffer is acetic acid (CH3COOH) and sodium acetate (CH3COONa). A common weak base buffer is made of ammonia (NH3) and ammonium chloride (NH4Cl).
What are the 3 buffer systems in the body?
The body’s chemical buffer system consists of three individual buffers: the carbonate/carbonic acid buffer, the phosphate buffer and the buffering of plasma proteins.
What are the 4 buffer systems in the body?
There are several buffer systems in the body. The most important include: (1) bicarbonate buffer (HCO3–/CO2), (2) haemoglobin buffer (in erythrocytes), (3) phosphate buffer, (4) proteins, and (5) ammonium buffer.
What is a buffer system and how does it work?
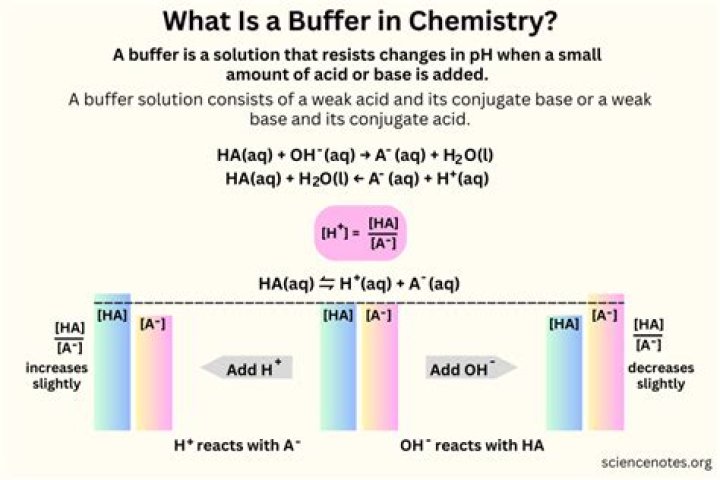
A buffer is a solution that can resist pH change upon the addition of an acidic or basic components. It is able to neutralize small amounts of added acid or base, thus maintaining the pH of the solution relatively stable. This is important for processes and/or reactions which require specific and stable pH ranges.
What is the most common buffer?
carbonic acid bicarbonate buffer
The body’s chemical buffer system consists of three individual buffers out of which the carbonic acid bicarbonate buffer is the most important. Cellular respiration produces carbon dioxide as a waste product. This is immediately converted to bicarbonate ion in the blood.
What are common buffers?
Common buffer compounds used in biology
| Common name (chemical name) | pKa, 25 °C | Mol. weight |
|---|---|---|
| TAPS, ([tris(hydroxymethyl)methylamino]propanesulfonic acid) | 8.43 | 243.3 |
| Bicine, (2-(bis(2-hydroxyethyl)amino)acetic acid) | 8.35 | 163.2 |
| Tris, (tris(hydroxymethyl)aminomethane, or 2-amino-2-(hydroxymethyl)propane-1,3-diol) | 8.07 | 121.14 |
What is protein buffer system?
PROTEIN BUFFER SYSTEM. Protein buffer system helps to maintain acidity in and around the cells. Haemoglobin makes an excellent buffer by binding to small amounts of acids in the blood, before they can alter the pH of the blood. Other proteins containing amino acid histidine are also good at buffering.
What is the most important buffer system in the body?
Carbonic-Acid-Bicarbonate Buffer
The Carbonic-Acid-Bicarbonate Buffer in the Blood By far the most important buffer for maintaining acid-base balance in the blood is the carbonic acid-bicarbonate buffer.
What are the components of a buffer system?
A buffer solution is made up of a weak acid and its conjugate base or a weak base and its conjugate acid. The two components maintain a pH balance that resists change when strong acids or bases are added to it.
What do a buffer consist of?
A buffer consists of a weak acid and its conjugate base or a weak base and its conjugate acid. Buffer capacity is the amount of acid or base that can be added before the pH of a buffer changes. An example of a buffer solution is bicarbonate in blood, which maintains the body’s internal pH.
What is buffer and its types?
Buffers are broadly divided into two types – acidic and alkaline buffer solutions. Acidic buffers are solutions that have a pH below 7 and contain a weak acid and one of its salts. Alkaline buffers, on the other hand, have a pH above 7 and contain a weak base and one of its salts.