What are the examples of amides?
What are the examples of amides?
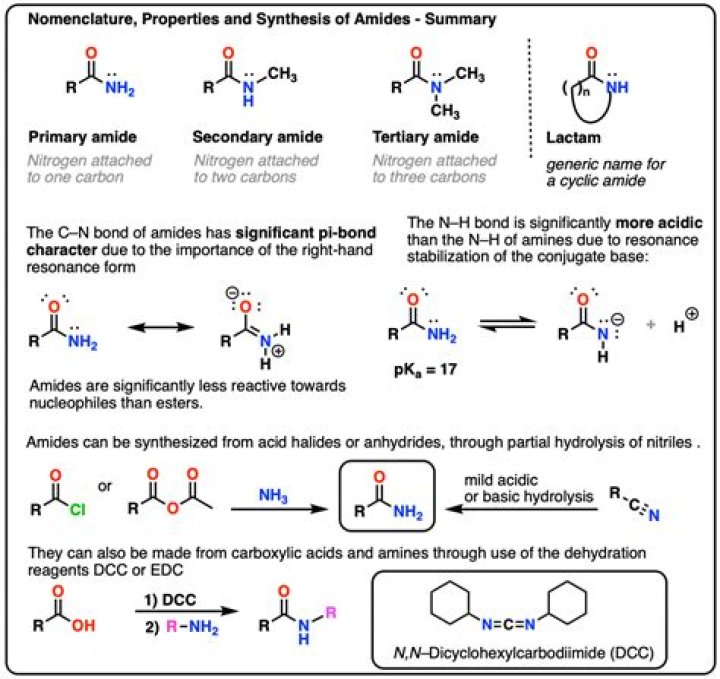
Common examples of amides are acetamide H3C–CONH2, benzamide C6H5–CONH2, and dimethylformamide HCON(–CH3)2. Amides are qualified as primary, secondary, and tertiary according to whether the amine subgroup has the form –NH2, –NHR, or –NRR’, where R and R’ are groups other than hydrogen.
Why are amides used in drugs?
Local anesthetics are used for local pain relief. Local infiltration of lidocaine or bupivacaine at the base of the involved digits decreases sympathomimetic input, reduces ischemic pain, and improves blood flow.
Where can amide be found?
There are no practical natural sources of simple covalent amides, although polyamides (amides linked together to form large molecules called polymers) occur in great abundance as the protein of living systems. Simple amides ordinarily are prepared by reaction of acids or acid halides with ammonia or amines.
Are amides explosive?
Safety. Sodium amide reacts violently with water to produce ammonia and sodium hydroxide and will burn in air to give oxides of sodium and nitrogen dioxide. In the presence of limited quantities of air and moisture, such as in a poorly closed container, explosive mixtures of peroxides may form.
How are amides named?
Primary amides are named by changing the name of the acid by dropping the -oic acid or -ic acid endings and adding -amide. It is not necessary to include the location number in the name because it is assumed that the functional group will be on the end of the parent chain.
How are amides made?
Amides are most commonly prepared though the reaction of an acid chloride with ammonia, a 1o amine, or 2o amine. In an analogous reaction, an amide can be prepared through the reaction of a carboxylic acid and an amine using a coupling agent such as DCC.
What drugs contain amides?
The amides commonly used include lidocaine, mepivacaine, prilocaine, bupivacaine, and articaine.
Is procaine a drug?
Procaine is a local anesthetic drug of the amino ester group. It is most commonly used in dental procedures to numb the area around a tooth and is also used to reduce the pain of intramuscular injection of penicillin.
How is an amide formed?
Amides generally are formed from acid chlorides, acid azides, acid anhydrides, and esters. It is not practical to prepare them directly from an amine and a carboxylic acid without strong heating or unless the reaction is coupled to a second reaction that “activates” the acid.
Is Penicillin an amide?
Structure. There are many types of penicillin, all of which belong to a larger family of antibiotics called β-lactams. The name is derived from their central ring structure – a 4-membered ring consisting of a cyclic amide called a lactam.
Are amides dangerous?
* Lithium Amide is a HIGHLY FLAMMABLE and REACTIVE chemical and a DANGEROUS FIRE and EXPLOSION HAZARD.
Are amides safe?
The Panel concluded that amino acid alkyl amides are safe in the present practices of use and concentration in cosmetics, when formulated to be nonirritating.
What are the two main uses of amides?
Uses of Amides 1 Amides are used for the preparation of primary amines. 2 Certain amides such as dimethylformamide (DMF) and dimethylacetamide (DMA) are used as solvents for polar and non- polar… More …
What’s the difference between amides and amines in chemistry?
Amides are molecules that contain nitrogen atoms connected to the carbon atom of a carbonyl group. Like amines, various nomenclature rules may be used to name amides, but all include use of the class-specific suffix –amide:
Where does the name amide come from in chemistry?
Updated March 05, 2019. An amide is a functional group containing a carbonyl group linked to a nitrogen atom or any compound containing the amide functional group. Amides are derived from carboxylic acid and an amine. Amide is also the name for the inorganic anion NH 2.
How are simple amides produced in the body?
Simple amides ordinarily are prepared by reaction of acids or acid halides with ammonia or amines. They can also be produced by the reaction of water with nitriles. Get a Britannica Premium subscription and gain access to exclusive content.
What is an example of an amide?
Examples of amides include carboxamides, sulfonamides, and phosphoramides. Nylon is a polyamide . Several drugs are amides, including LCD, penicillin, and paracetamol .
Is amide an acid or base?
An amide (/ˈæmaɪd/ or /ˈæmɪd/ or /ˈeɪmaɪd/), also known as an acid amide, is a compound with the functional group R nE(O) xNR′ 2 (R and R′ refer to H or organic groups).
What is primary amide?
Primary Amide. A primary (1°) amide is an amide in whose molecule the nitrogen atom is bonded to only one carbon atom.
What is the pKa of amide?
Amide properties. Compared to amines, amides are very weak bases. While the conjugate acid of an amine has a pKa of about 9.5, the conjugate acid of an amide has a pKa around -0.5.