What conformation must the diene be to participate in a Diels-Alder reaction?
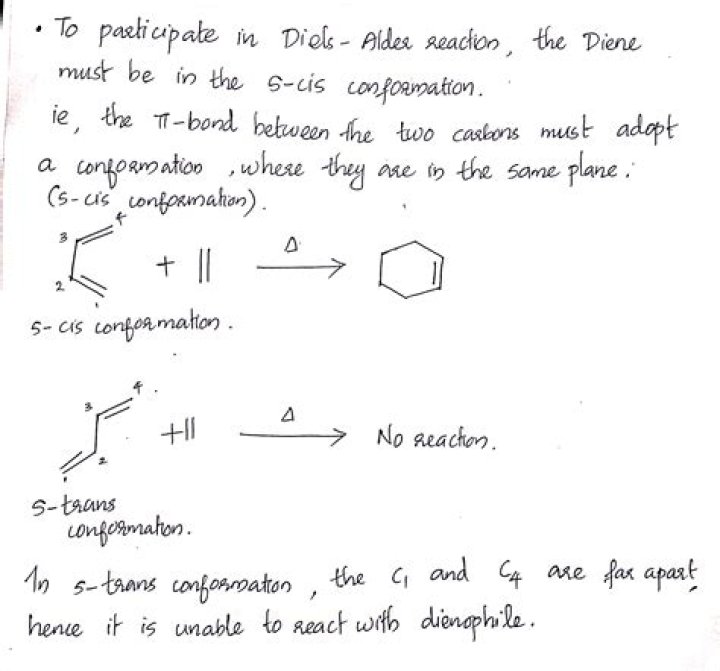
What conformation must the diene be to participate in a Diels-Alder reaction?
s-cis conformation
The Diene Must Be In The s-cis conformation. Substituents Do NOT Change the pattern of bonds formed/bonds broken… … but they do affect the rate!
What makes a good diene for Diels-Alder?
A particularly rapid Diels-Alder reaction takes place between cyclopentadiene and maleic anhydride. We already know that cyclopentadiene is a good diene because of its inherent s-cis conformation. Notice that the both of these reactions require compounds in which two double bonds are separated by three single bonds.
Which factors govern the Diels-Alder reaction?
This means that three factors influence the Diels- Alder reaction, namely, the electron density of the carbons taking part in the addition reaction, the frontier orbital energy levels of the reactants, and, the steric energy of the transition state and the end product.
What dienes can undergo the Diels-Alder reaction?
Only conjugated dienes in the s-cis conformation can undergo Diels-Alder reactions. Dienes that are located in rings can’t rotate, and so are stuck in either an s-cis or s-trans conformation.
What are Dienes and Dienophiles?
As nouns the difference between diene and dienophile is that diene is (organic chemistry) an organic compound, especially a hydrocarbon, containing two double bonds while dienophile is (organic chemistry) a compound that readily reacts with a diene; especially an alkene in the diels-alder reaction.
What makes diene more reactive?
Remember that electron-donating groups increase the reactivity of the diene: Therefore, electron-donating groups on the diene increase its reactivity, while electron-withdrawing groups on the dienophile lower the LUMO energy level, thus support this electron flow as well.
How do you rank up diene reactivity?
Dienophile reactivity is increased by: (A) electron-withdrawing substituents, (B) a weak π-bond, and (C) release of ring strain.
What are the requirements for the dienes and the Dienophiles?
(ii) The diene component must be able to assume a s-cis conformation. (iii) Electron withdrawing groups on the dienophile facilitate reaction. (iv) Electron donating groups on the diene facilitate reaction. (v) Steric hindrance at the bonding sites may inhibit or prevent reaction.
Which dienes is not capable of participate in a Diels-Alder reaction?
The s-cis conformation is higher in energy than the s-trans conformation, due to steric hindrance. For some dienes, extreme steric hindrance causes the s-cis conformation to be highly strained, and for this reason such dienes do not readily undergo Diels-Alder reactions.
Which dienes Cannot undergo Diels-Alder reaction?
Diels-Alder reaction occurs between a diene (a compound consisting of the double bond in conjugation) and a dienophile. The given molecule is a conjugated diene but is a rigid molecule; thus, it cannot undergo the Diels-Alder reaction.



