What distills first from a mixture of acetone and water
In the distillation method, the two liquids present in the distillation flask are boiled and the liquid with lower boiling point, that is the acetone, vaporises first and gets condensed and isolated from the mixture, leaving behind the water in the flask.
What happens when you mix water with acetone?
When acetone is mixed with water it completely dissolves in it. In this type of reaction acetone is usually the solute and water is the solvent. When these two compounds are mixed there is a formation of hydrogen bonds resulting in a homogeneous solution.
How will you separate acetone and water by simple distillation?
Difference in the boiling points of acetone and water is 25K. The mixture of water and acetone is taken in a distillation flask and is slowly heated till acetone evaporates. Water is collected in the distillation flask and acteone vapours are condensed to get acetone. Thus the mixture is separated.
Which liquid distilled first water or alcohol?
Ethanol has a lower boiling point than water, so it evaporates first. The ethanol vapour is then cooled and condensed inside the condenser to form a pure liquid known as the distillate.How do you distill acetone?
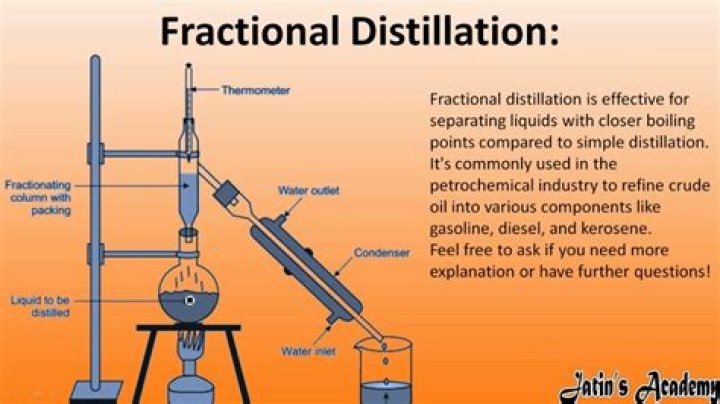
Acetone can be distilled from water while keeping a close eye on the temperature of the solution. Best possible method is fractional distillation at controlled temperature . set the distillation system temperature to boiling point of acetone then it willbe carried over which can be cooled later.
Why does acetone and water not mix?
Water is much more polar than acetone though – think of acetone as being intermediate in polarity between water, and say, hexanes. Acetone is still non-polar enough to dissolve non-polar compounds that do not dissolve in water.
Is acetone soluble in water?
The slightly positive charge on each hydrogen can attract slightly negative oxygen atoms on other water molecules, forming hydrogen bonds. If acetone is added to water, acetone would completely dissolve.
How do you separate methyl alcohol and acetone?
Thus, the acetone and methanol can be separated by the fractional distillation. Hence, (B) is the correct option.How do you separate isopropanol from water?
Abstract Ethanol, isopropanol and water cannot be separated from each other by distillation or rectification because of minimum azeotropes. They are readily separated by extractive distillation.
What mixtures can be separated by distillation?Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or in the separation of two or more liquids having different boiling points, as in the separation of gasoline, kerosene, and lubricating oil from crude oil.
Article first time published onWhich of the following technique is used for separation of acetone from water?
Fractional distillation is used for the purification of water as well as separating acetone and water.
Can you distill acetone from water?
That is, the distillation process. In the distillation method, the two liquids present in the distillation flask are boiled and the liquid with lower boiling point, that is the acetone, vaporises first and gets condensed and isolated from the mixture, leaving behind the water in the flask.
Which distillation setup gives the best separation of acetone and water Why?
The reason that fractional distillation gives better separation between the liquids is because the glass beads in the fractionating column provide “theoretical plates” on which the refluxing liquid can condense, re-evaporate, and condense again, essentially distilling the compound over and over.
Does acetone form an azeotrope with water?
Acetone and water are miscible in all proportions but, somewhat surprisingly, do not interact to form a binary azeotrope. The boiling point of acetone is ca 56 C and will have a significanf partial vapour pressure when the mixture with water is heated to 40 C.
What common materials are soluble in acetone?
Acetone is used to dissolve other chemical substances and mixes readily with water, alcohol, dimethylformamide, chloroform, ether and most oils.
Would acetone and water make a good solvent pair for a liquid liquid extraction Why or why not?
The more polar the organic solvent, the more it is miscible (soluble) with water. For example, polar solvents such as methanol, ethanol and acetone are miscible with water, thus not suitable for liquid-liquid extraction.
Is acetone and water miscible or immiscible?
Acetone and Water are miscible, and water and diethyl ether are not.
Is acetone soluble in alcohol?
NamesBoiling point56.05 °C (132.89 °F; 329.20 K)Solubility in waterMiscibleSolubilityMiscible in benzene, diethyl ether, methanol, chloroform, ethanollog P−0.16
What can you not mix with acetone?
You can also form chloroform by mixing acetone with bleach. Acetone is commonly found in nail polish remover and in certain paint or varnish removers. Ammonia and bleach: This combination is dangerous, producing vapors that can cause severe damage to your respiratory system.
How do you remove water from acetone?
- Fractional distillation. Gently heat the mixture and recover the acetone vapor with a condenser. …
- Add a desiccant to the mixture to absorb the water, then filter off the acetone. Plaster of Paris will do the job and is cheap and easy to obtain.
How do you distill ethanol from water?
For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points. When the mixture is heated, one liquid evaporates before the other.
How would you separate sand and water into their components?
It is easy to separate sand and water by filtering the mixture. Salt can be separated from a solution through evaporation. The water can also be recovered as well as the salt if the water vapour is trapped and cooled to condense the water vapour back into a liquid. This process is called distillation.
How is ethanol obtained from the fermentation mixture?
The fermentation mixture contains yeast cells and insoluble substances. These are separated from the impure ethanol solution by filtration . Fractional distillation is then used to produce a concentrated solution of ethanol from the filtrate . … When the mixture is heated, ethanol evaporates more readily.
How will you separate a mixture of acetone and alcohol class 9?
A mixture of acetone and alcohol can be separated by fractional distillation.
Can acetone and methanol be separated by simple distillation?
The boiling points of acetone and methanol are 56ºC and 64.7ºC respectively which concludes that the difference in their boiling points is not greater than 25ºC and hence cannot be separated by simple distillation.
How will you separate ethanol from acetone mention the reason for choosing the method?
The boiling point of acetone is 560C and that of ethyl alcohol is 78.40C. The mixture of acetone and ethanol can be separated using fractional distillation. er are two immiscible liquids, thus their mixture can be separated using separating funnel.
What is the first step in the process of distillation?
The process of distillation begins with heating a liquid to boiling point. The liquid evaporates, forming a vapor. The vapor is then cooled, usually by passing it through pipes or tubes at a lower temperature. The cooled vapor then condenses, forming a distillate.
Can you separate lemon juice from water?
Lemonade is a special type of mixture called a solution. The ingredients in a solution cannot be separated by hand because of changes in the ingredients’ physical properties.
What are 5 ways to separate a mixture?
- chromatography: Involves solvent separation on a solid medium.
- distillation: Takes advantage of differences in boiling points.
- evaporation: Removes a liquid from a solution to leave a solid material.
- filtration: Separates solids of different sizes. References.
Which technique is used to separate acetone and benzene by the following method?
A mixture of acetone and benzene can be separated by fractional distillation.
Which method is used to separate the mixture of acetone and benzene?
Answer: They could be separated by fractional distillation since acetone have b.p of 329k and benzene has b.p of 353k. Since b.p difference is less than 25k. so they are separated by fractional distillation.