What factors affect the melting point of an ionic compound
The charge on the ions.Size of the ions.
What are the factors affecting the melting point?
Molecular composition, force of attraction and the presence of impurities can all affect the melting point of substances.
What factors affect melting and boiling point?
Just like with boiling points, the presence of polar and hydrogen-bonding groups on organic compounds generally leads to higher melting points. The size of a molecule influences its melting point as well as its boiling point, again due to increased van der Waals interactions between molecules.
What causes ionic compounds to have high melting points?
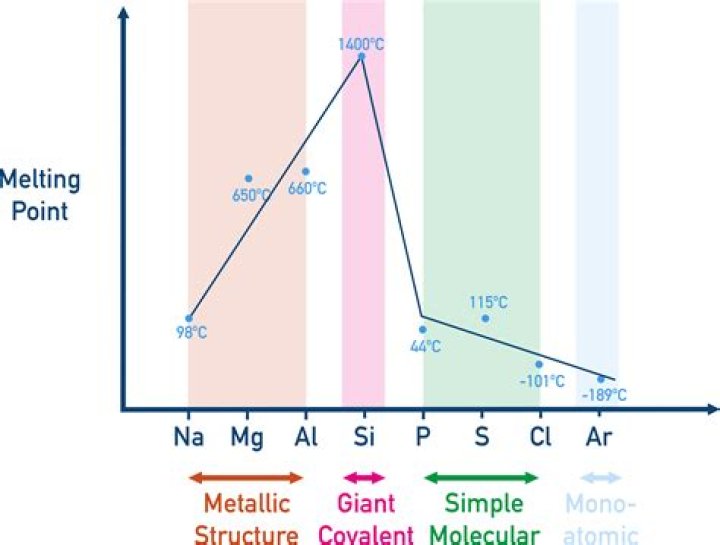
Ionic compounds are held together by strong electrostatic forces of attraction between oppositely charged ions. These compounds have high melting points and high boiling points because of the large amounts of energy needed to break the many strong bonds. … When atoms share pairs of electrons, they form covalent bonds.How does ionic bonding affect melting point?
Short answer: Compounds with ionic bonding have higher melting points than those with covalent bonding. Intermolecular forces determine the melting points of compounds. … It takes much more energy to separate ions than it does to separate molecules from each other.
Do ionic compounds melt easily?
Ionic compounds typically have much higher melting points than molecular compounds. … To melt a molecular substance, you need to break these weak intermolecular forces between neutral molecules, which is why ionic compounds generally have much higher melting points than molecular compounds.
What causes high melting point?
High melting point metals have strong intermolecular forces between atoms. Electrostatic attraction forces between metal ions and free electrons create strong metallic bonds with stronger bonds resulting in higher melting temperatures.
What is the melting point of ionic compounds?
Ionic compoundMelting pointBoiling pointNaCl801°C1,413°CMgO2,852°C3,600°CHow does intermolecular forces affect melting point?
The stronger the intermolecular forces are, the more energy is required, so the higher the melting point is. Many intermolecular forces depend on how strongly atoms in the molecule attract electrons — or their electronegativity. … Hydrogen bonds are among the strongest intermolecular forces.
Do ionic compounds have higher melting points than metals?Both metals and ionic solids are non-molecular materials, that are held together by strong electrostatic forces. Because metallic bonding is rather fluid, i.e. bonding results from the delocalization of valence electrons across the metallic lattice, metals tend to have lower melting points.
Article first time published onWhat determines the melting point of a compound?
The force of attraction between the molecules affects the melting point of a compound. Stronger intermolecular interactions result in higher melting points. … In organic compounds the presence of polarity, or especially hydrogen bonding, generally leads to higher melting point.
How does symmetry affect melting point?
Molecular symmetry has a pronounced effect on the melting properties and solubility of organic compounds. As a general rule, symmetrical molecules in crystalline form have higher melting temperatures and exhibit lower solubilities compared with molecules of similar structure but with lower symmetry.
What causes a melting point to be lower than the literature value?
The melting points of compounds may be lower than the reported values because it may contain small amounts of the impurities or solvents. Impurities in a solid cause a melting point depression because the impurity disrupts the crystal lattice energies.
Why are the melting points of ionic compounds higher than covalent quizlet?
Why? Ionic compounds have high melting points and boiling points because the electrostatic forces of attraction between oppositely charged ions are strong and large amount of energy is require to separate the ions.
Why are ionic compounds usually high melting point Whilst most simple covalent compound have low melting points explain the high melting point of diamond?
Key Points Ionic compounds are formed from strong electrostatic interactions between ions, which result in higher melting points and electrical conductivity compared to covalent compounds. Covalent compounds have bonds where electrons are shared between atoms.
How do the melting points of ionic compounds compare to covalent compounds?
Examples include sugar crystals and diamond. Melting and boiling points: Ionic compounds tend to have higher melting and boiling points than covalent compounds. … This is because covalent compounds dissolve into molecules while ionic compounds dissolve into ions, which can conduct charge.
Does electronegativity affect melting point?
As you must be knowing, opposites attract and the sheer electrostatic attraction between the molecules of the compound will tend to make the compound, as a whole, have a much higher Boiling and/or Melting point. Higher electronegativity means higher polarity in the molecule containing such atom.
Which of the following properties affects the melting and boiling points of molecules?
The size of the melting or boiling point will depend on the strength of the intermolecular forces. The presence of hydrogen bonding will lift the melting and boiling points. The larger the molecule the more van der Waals attractions are possible – and those will also need more energy to break.
Which intermolecular force would affect melting point the least?
Type of compoundIntermolecular forces presentRelative order of boiling and melting pointsNonpolar covalent compoundsLondon dispersion forces4, lowest
How do melting points compare to ionic compounds?
To compare 2 ionic compounds’ melting point, there are generally 3 determinants: Ionic Radii, smaller the ionic radius, closer the ions are to each other, therefore stronger the electrostatic attraction between them.
What causes a melting point to be higher than the literature value?
Inappropriate sample size for the apparatus; poor placement of the sample with respect to the thermometer, heating too fast, not calibrated thermometer or instrument, the material you are measuring is NOT what you think it is, finally, if you are the perfect lab person, the literature value is incorrect.
Does branching affect melting point?
It’s a nice story: branching decreases melting point and boiling point. … Starting with the simplest branched compound, as you increase branching, you will increase the melting point, but decrease the boiling point.
Why does symmetry increase melting point?
Symmetry and Melting Point Aside from the intermolecular interactions, however, the melting point depends also on how the molecules are packed or arranged in the solid form. The more symmetrical they are, the better they pack and form a perfect crystal lattice which results in a higher melting point.
What is the effect of branching on melting point of alkanes?
Due to branching the surface area of molecules decreases which makes it more compact, it will be easier to pack them tightly and it will be hard to break that compact structure so this explains that due to branching the melting point increases.
How does purity affect melting point?
A substance (solid) containing soluble impurities usually melts at a lower temperature than the pure compound. It can also melt over a wide range of temperatures and is called the “melting point depression.” In general, the smaller the range of melting temperatures, the higher the purity of the sample.
What can lower melting point?
The presence of even a small amount of impurity will lower a compound’s melting point by a few degrees and broaden the melting point temperature range. Because the impurity causes defects in the crystalline lattice, it is easier to overcome the intermolecular interactions between the molecules.
Why do impurities affect melting point?
Foreign substances in a crystalline solid disrupt the repeating pattern of forces that holds the solid together. Therefore, a smaller amount of energy is required to melt the part of the solid surrounding the impurity. This explains the melting point depression (lowering) observed from impure solids.
How does the melting point of ionic compounds compare to that of covalent compounds quizlet?
Ionic compounds have higher melting points than covalent compounds. Ionic compounds are composed of a metal and a nonmetal while covalent compounds are composed of all nonmetals. … Solid ionic compounds do not conduct electricity.
How do ionic bonds affect the properties of ionic compounds?
How do ionic bonds affect the properties of ionic compounds? The bonds prevent electrons from moving throughout the crystal, so a solid ionic compound is a poor conductor. … One compound is covalent, and the other is ionic.
Does charge affect melting point?
In general, the greater the charge, the greater the electrostatic attraction, the stronger the ionic bond, the higher the melting point.