What is an allylic positive charge?
What is an allylic positive charge?
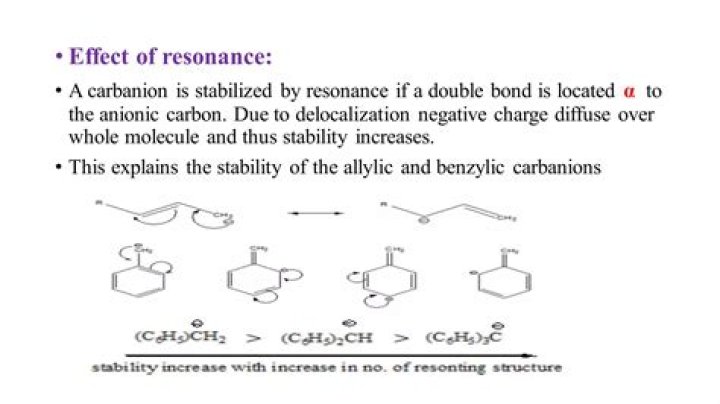
Allylic carbocations are a common conjugated system. The positive charge of a carbocation is contained in a P orbital of a sp2 hybrizied carbon. This allows for overlap with double bonds. The positive charge is more stable because it is spread over 2 carbons.
What is a primary allylic carbocation?
A primary allylic carbocation is an allylic carbocation in each resonance form of which the formal charge of +1 is on a primary carbon.
Is allylic or tertiary carbocation?
If in one or both resonance forms of an allylic carbocation the formal charge of +1 is on a tertiary carbon, the allylic carbocation is called a tertiary (3°) allylic carbocation.
What is carbocation Slideshare?
A carbocation is an organic molecule, an intermediate, that forms as a result of the loss of two valence electrons, normally shared electrons, from a carbon atom that already has four bonds. This leads to the formation of a carbon atom bearing a positive charge and three bonds instead of four.
What is carbocation explain its structure and stability?
A carbocation is a molecule in which a carbon atom has a positive charge and three bonds. The carbon has 6 electrons in its valence shell. Due to this, it is an electron-deficient species, also known as an electrophile.
Which is an allylic carbon?
An allylic carbon is a carbon atom bonded to a carbon atom that in turn is doubly bonded to another carbon atom.
Which is more stable allylic carbocation or tertiary carbocation?
Allylic Carbocation is more stable due to Greater mesomeric effect where as on the other side tertiary butyl Carbocation is stabilized by triple inductive effect.
Why is the allylic cation different in stability compared to a primary cation?
Allylic carbocations are a common conjugated system. This delocalization stablizes the allyl carbocation making it more stable than a normal primary carbocation. The positive charge of a carbocation is contained in a p orbital of a sp 2 hybridized carbon. This allows for overlap with double bonds.
Is allylic carbocation or 3rd carbocation more stable?
tert butyl carbocation is 3 degree and allylic carbocation is 1 degree thus tert is more stable.