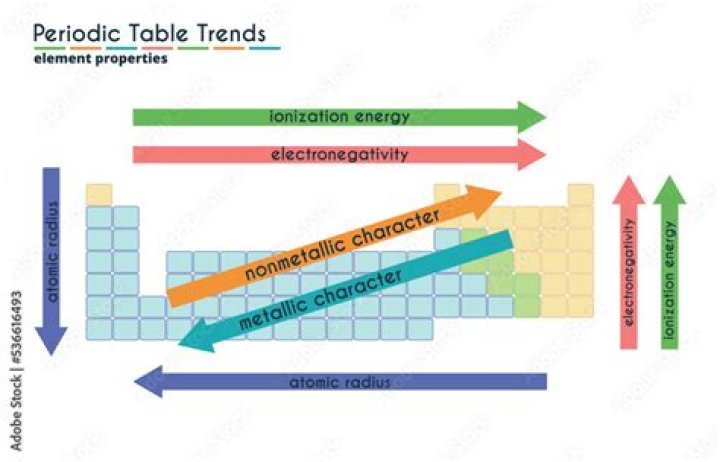
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties..
In this regard, what does periodic trend mean?
A periodic trend is a regular variation the properties of an element with increasing atomic number. A periodic trend is attributed to regular variations in the atomic structure of each element.
Additionally, what can a periodic trend predict about an element? 1 Answer. The Periodic Table can predict the properties of new elements, because it organizes the elements according to their atomic numbers. Creating new elements is not a simple process. Scientists use a particle accelerator to smash light atoms into a thin metallic foil that contains heavier atoms.
Keeping this in view, what is the general periodic trend for valence electrons?
The general trend when going left to right across a period is that the electronegativity increases. This is due primarily to the fact discussed in the ionization energy that electrons with more full valence shells tend to attract electrons, and elements with less full shells tend to give away electrons.
Is Melting Point A periodic property?
Melting points and boiling points show periodic properties. This means that they vary in a regular way or pattern depending on their position in the Periodic Table.
Related Question Answers
What are the 3 periodic trends?
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character.What are the trends in modern periodic table?
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an element's properties.What causes electronegativity?
Electronegativity increases as you move across the periodic table from left to right. This occurs due to a greater charge on the nucleus, causing the electron bonding pairs to be very attracted to atoms placed further right on the periodic table. Fluorine is the most electronegative element.What is the trend in ionization energy across a period?
The general trend is for ionization energy to increase moving from left to right across an element period. Moving left to right across a period, atomic radius decreases, so electrons are more attracted to the (closer) nucleus.Is density a periodic property?
Density Is a Periodic Property. Dmitri Mendeleev proposed the periodic law for the classification of elements in 1869-1871. After observing trends in the properties of elements when they were arranged in order of increasing atomic mass, Mendeleev made a startling prediction.How do you determine electronegativity?
To calculate electronegativity, start by going online to find an electronegativity table. You can then assess the quality of a bond between 2 atoms by looking up their electronegativities on the table and subtracting the smaller one from the larger one. If the difference is less than 0.5, the bond is nonpolar covalent.What is the trend of electronegativity in the periodic table?
So, as you move down a group on the periodic table, the electronegativity of an element decreases because the increased number of energy levels puts the outer electrons very far away from the pull of the nucleus. Electronegativity increases as you move from left to right across a period on the periodic table.What is periodic property?
The phenomenon in which repetition of similar properties of elements occurs or reappears at atomic number intervals of 2,8,8,18,18 ,32 is called periodicity and these properties are called periodic properties. These include atomic radii, Ionisation potential, Electronegativity,Electron affinity etc.What is the most electronegative element?
Electronegativity varies in a predictable way across the periodic table. Electronegativity increases from bottom to top in groups, and increases from left to right across periods. Thus, fluorine is the most electronegative element, while francium is one of the least electronegative.What is ionization energy in periodic table?
Ionization energy refers to the amount of energy needed to remove an electron from an atom. Ionization energy decreases as we go down a group. Ionization energy increases from left to right across the periodic table.What are group trends?
Group Trend: as you go down a column, atomic radius increases. Periodic Trend: as you go across a period, (L to R), atomic radius decreases. Ionization Energy Trends. Group Trend: as you go down a column, ionization energy decreases. Periodic Trend: as you go across a period, ionization energy increases.What is a period number on the periodic table?
Periods in the periodic table. In each period (horizontal row), the atomic numbers increase from left to right. The periods are numbered 1 through 7 on the left-hand side of the table. Elements that are in the same period have chemical properties that are not all that similar.How do you arrange ionization energy?
The first ionization energy varies in a predictable way across the periodic table. The ionization energy decreases from top to bottom in groups, and increases from left to right across a period. Thus, helium has the largest first ionization energy, while francium has one of the lowest.Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).How do you make a periodic table?
Read the periodic table from top left to bottom right. The elements are ordered by their atomic numbers, which increase as you move across and down the periodic table. The atomic number is how many protons the element's atom possesses.What are the elements of a trend?
You now understand the three fundamental elements of a trend: basic human needs; change (both longer-term shifts and short term triggers); innovations and can identify points of tension and emerging customer expectations, which are where the key opportunities lie when it comes to consumer trends.What happens when you go down the periodic table?
The reactivity all increases as you go down the periodic table, for example rubidium is far more reactive than sodium. Electronegativity: This property determines how much the element attracts electrons. The electronegativity increases going from left to right, and it decreases going down the table.Why is electronegativity a periodic trend?
The electronegativity of atoms increases as you move from left to right across a period in the periodic table. This is because as you go from left to right across a period, the nuclear charge is increasing faster than the electron shielding, so the attraction that the atoms have for the valence electrons increases.What happens as you go down the periodic table?
Moving down a group in the periodic table, the number of filled electron shells increases. In a group, the valence electrons keep the same effective nuclear charge, but now the orbitals are farther from the nucleus. A cation is an atom that has lost one of its outer electrons.