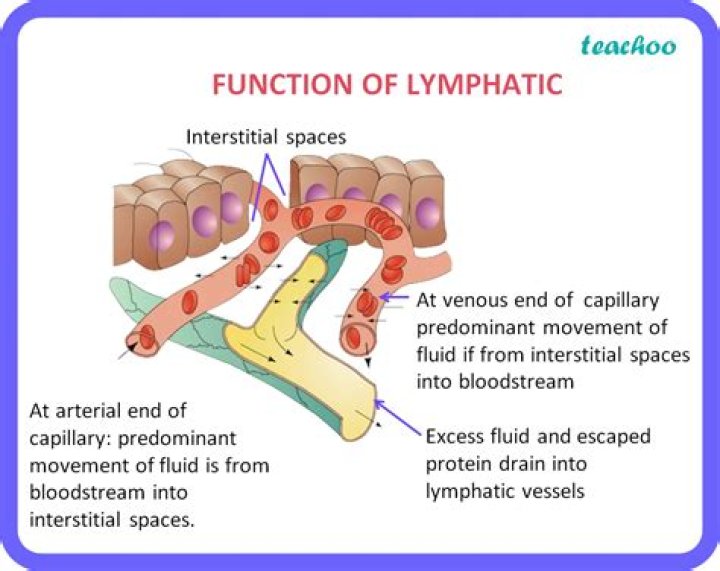
What is called functional group
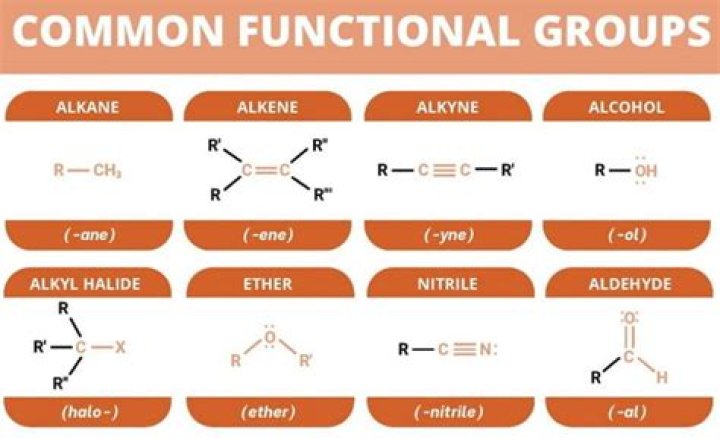
A functional group is a group of atoms in a molecule with distinctive chemical properties, regardless of the other atoms in the molecule. The atoms in a functional group are linked to each other and to the rest of the molecule by covalent bonds.
What do you mean by functional group?
In organic chemistry, a functional group is a specific group of atoms or bonds within a compound that is responsible for the characteristic chemical reactions of that compound. … The atoms of a functional group are linked together and to the rest of the compound by covalent bonds.
What are called functional groups give example?
An atom/group of atoms joined in a specific manner which is responsible for the characteristics chemical properties of the organic compounds is called a functional group. Examples are hydroxyl group (-OH), aldehyde group(-CHO), Ketonic group (-CO-),Carboxlic acid group(-COOH) etc.
What is called functional group Class 10?
The atom or the group of atoms by which the characteristic reactions of organic compounds are determined, that atom or group of atoms is called the functional group.What is functional group in Brainly?
The functional group is defined as an atom or group of atoms joined in a specific manner, which gives the chemical properties of the organic compound and are the centers for chemical reactivity. Compounds having a similar functional group have undergone similar reactions.
What is functional group class 11th?
A functional group may be defined as as atom or a group of atoms present in a molecule which largely determines its chemical properties. … All the organic compounds containing the same functional group show similar chemical reactions.
What are the 4 functional groups?
Some of the important functional groups in biological molecules include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl groups. These groups play an important role in the formation of molecules like DNA, proteins, carbohydrates, and lipids.
What are functional groups Brainly 10?
Answer: Functional groups are the atoms or group of atoms attached to a hydrocarbon chain which changes the property of parent hydroarbon chain .What is functional group in chemistry class 11?
What is called a functional group? Functional groups are groups of one or more atoms with distinctive chemical properties regardless of what is attached to them. The atoms of functional groups are bound by covalent bonds with one another and with the rest of the molecule.
What are functional groups in biology?A functional group is a specific group of atoms within a molecule that is responsible for a characteristic of that molecule. Many biologically active molecules contain one or more functional groups.
Article first time published onWhat is a functional group give two examples of functional group?
Chemical classGroupExampleAlcoholHydroxylMethanolKetoneCarbonylButanone (Methyl ethyl ketone)AldehydeAldehydeAcetaldehyde (Ethanal)Acyl halideHaloformylAcetyl chloride (Ethanoyl chloride)
Is the phosphate group Polar?
The phosphate group is the negatively-charged polar head, which is hydrophilic. The fatty acid chains are the uncharged, nonpolar tails, which are hydrophobic. Since the tails are hydrophobic, they face the inside, away from the water and meet in the inner region of the membrane.
What are the 7 functional groups?
Functional groups include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl.
Is ch3 a methyl?
A methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms — CH3. In formulas, the group is often abbreviated Me. … It is a very stable group in most molecules.
What functional group is basic?
Functional GroupPropertiesAminoCharged (forms R-NH 3+start subscript, 3, end subscript, start superscript, plus, end superscript) at the pH of most biological systems. Since amino groups can remove H +start superscript, plus, end superscript from solution, they are considered basic.
What is functional group isomerism?
Functional isomerism occurs when substances have the same molecular formula but different functional groups. This means that functional isomers belong to different homologous series. … There are three functional group isomers of which you need to be aware: alcohols and ethers. aldehydes and ketones.
How many functional groups are there?
Functional groups include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl.
What is the functional group order?
- CARBOXYLIC ACIDS (highest priority among carbon-containing functional groups).
- CARBOXYLIC ACID DERIVATIVES.
- OTHER GROUPS CONTAINING OXYGEN OR NITROGEN.
- ALKENES AND ALKYNES. …
- LOWEST PRIORITY.
What is the functional group of amides?
Amides are compounds that consist of a carbonyl functional group which is connected to both an amine group and a hydrocarbon group (or hydrogen atom). A carbonyl functional group consists of a carbon atom which is double-bonded with an oxygen atom.
What is functional group of aldehyde?
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.
Is NH a functional group?
In organic chemistry, the most common functional groups are carbonyls (C=O), alcohols (-OH), carboxylic acids (CO2H), esters (CO2R), and amines (NH2). It is important to be able to recognize the functional groups and the physical and chemical properties that they afford compounds.
What is functional group explain with example class 10?
The atom or the group of atoms by which the characteristic reactions of organic compounds are determined, that atom or group of atoms is called the functional group. Some organic functional groups are as follows: Carboxylic acid: −COOH.
What are functional groups in chemistry?
functional group, any of numerous combinations of atoms that form parts of chemical molecules, that undergo characteristic reactions themselves, and that in many cases influence the reactivity of the remainder of each molecule.
How do you identify a functional group?
Functional groups are the groups of atoms that are attached to the carbon backbone of organic molecules. Functional groups are responsible for the characteristic chemical reactions of organic compounds. They are less stable than the carbon backbone and are more likely to take part in chemical reactions.
What do carbonyl groups do?
Within biology, a carbonyl group within a molecule allows it to undergo the many reactions necessary to maintain life. Many common biological molecules contain a carbonyl group, which allows the cell the ability to create new molecules and modify the molecule with a number of other functional groups.
How do you name Esters?
Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.
What is meant by functional group in carbon compounds?
Functional group in a carbon compound may be defined as an atom or group of atoms or reactive part which is responsible for the characteristic properties of the compounds. The function group of ethanol(CH3CH2OH) is alcohol, i.e., −OH group.
What is polar functional group?
Functional groups can sometimes be classified as having polar or nonpolar properties depending on their atomic composition and organization. The term polar describes something that has a property that is not symmetric about it—it can have different poles (more or less of something at different places).
What is ketone functional group?
In chemistry, a ketone /ˈkiːtoʊn/ is a functional group with the structure R2C=O, where R can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond). … Many ketones are of great importance in biology and in industry.
Is a carbonyl group polar?
Thus, molecules containing the carbonyl group are polar. Compounds containing a carbonyl group have higher melting and boiling points than hydrocarbons containing the same number of carbon atoms and are more soluble in polar solvents such as water.
Is alcohol a functional group?
An alcohol is an organic compound with a hydroxyl (OH) functional group on an aliphatic carbon atom. Because OH is the functional group of all alcohols, we often represent alcohols by the general formula ROH, where R is an alkyl group. Alcohols are common in nature.