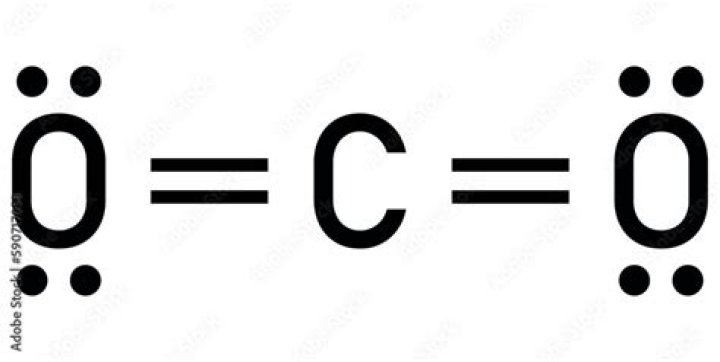Carbon (C) is the least electronegative atom in the CO2 Lewis structure and therefore should be placed at the center of the structure. The Lewis structure for CO2 has a total of 16 valence electrons. In order to complete the octets for all of the atoms in the structure you will need to form two double bonds..
Correspondingly, what is the shape of co2?
Carbon dioxide has two electron groups and no lone pairs. Carbon dioxide is therefore linear in electron-group geometry and in molecular geometry. The shape of CO2 is linear because there are no lone pairs affecting the orientation of the molecule.
Also Know, what is the structure of carbon dioxide? CO2
One may also ask, is co2 a Lewis acid or base?
Acids and Bases: Lewis Theory Carbon dioxide is a polar molecule whose positive center is on the carbon atom: This positive center is able to attract (and accept) the lone electron pairs present on the oxide ion (O2-). Thus, carbon dioxide is acting as a Lewis acid and the oxide ion is acting as a Lewis base.
Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.
Related Question Answers
What shape is h2s?
Answer and Explanation: The molecular shape of hydrogen sulfide is bent. The central atom sulfur is bonded to two hydrogen atoms.Is ch4 trigonal planar?
For example; four electron pairs are distributed in a tetrahedral shape. If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3).What is the molecular shape of CO?
The molecule is linear (only two atoms). Considering, now, the electron geometry, note that on each atom we only have two electron domains -- the triple bond, and the lone electron pair. Multiple bonds count as single electron domains.What is the structure of co2?
Carbon Dioxide (CO2) is a gas with a molecular structure composed of two oxygen atoms and one carbon atom. It is an important greenhouse gas because of its ability to absorb infrared wavelengths. Carbon dioxide is a chemical compound that is found in Earth's atmosphere as a gas.Is so2 linear or bent?
Carbon dioxide is linear, while sulphur dioxide is bent (V-shaped). In the carbon dioxide, the two double bonds try to get as far apart as possible, and so the molecule is linear. In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur.Is h2o linear or bent?
The oxygen has 6 valence electrons and thus needs 2 more electrons from 2 hydrogen atoms to complete its octet. The water molecule is bent molecular geometry because the lone electron pairs, although still exerting influence on the shape, are invisible when looking at molecular geometry.Is COCl2 trigonal planar?
The bond angle is 180o. arrangement - AX3 with “3 bonding pairs” & no lone pairs on the central atom). The three groups of electron pairs are arranged in a trigonal plane. Thus, the molecular shape of COCl2 is trigonal planar.Is HCN linear or bent?
Hydrogen Cyanide In this example, HCN, the Lewis diagram shows carbon at the center with no lone electron pairs. The carbon and nitrogen are bonded through a triple bond which counts as "one electron pair". Hence the molecule has two electron pairs and is linear.Is HCl a Lewis acid?
A Lewis acid is an electron -pair acceptor; a Lewis base is an electron-pair donor. An example is HCl vs H+: HCl is a classical acid, but not a Lewis acid; H+ is a Lewis acid when it forms an adduct with a Lewis base.What is Lewis acid give example?
Lewis Acids Examples include copper (Cu2), iron (Fe2+ and Fe3+), and hydrogen ion (H+). An atom, ion, or molecule with an incomplete octet of electrons can accept electrons. Examples include boron trifluoride (BF3) and aluminum fluoride (AlF3).Is calcium a Lewis acid?
Sodium, potassium, calcium and magnesium are common ions in biology. Because they attract electron donors, these ions are generally found in biological situations with water molecules stuck to them; remember that the oxygen atom in water is Lewis basic.Is bh3 Lewis acid?
By definition if any chemical species can accomodate electron from any other species then it is termed as a Lewis acid. A closer look will reveled that boron of BH3 is not completely filled up its octet. So it is able to uptake electrons from any electron rich speices. So BH3 can be regarded as a Lewis acid .Is AlCl3 a Lewis acid?
In AlCl3 it forms three bonds and hence outer shell has 6 electrons. Now Al needs two more electrons to complete its octet. By definition those which accepts electrons are called lewis acids. So AlCl3 is a Lewis acid.Why is co2 not a Lewis base?
CO2 is considered a Lewis acid. But CO2 can't accept electron pairs because oxygen and carbon both are electron sufficient (have complete octets). If the reason is within a lone pair of electrons on oxygen then ammonia (NH3), which also has a lone pair of electrons, should be acidic, too.Is SnCl2 a Lewis acid?
In SnCl2,the tin is electrin deficient centre. Due to presence of two chlorine atoms the electron deficiencyagain increases. Here sn centre acts as lewis acid. Again the chlorine centre is electron rich centre and hence it readily donates Electron to the electron deficient centre.Is BCl3 a Lewis acid?
The central atom in BCl3 (that is Boron, in trivalent state) has only six electrons around it — it is electron deficient and accepts electrons to complete it's octet. Therefore it acts as a Lewis acid by accepting electrons.What type of bond is co2?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, which is shown here as two lines and referred to as a double bond. When molecules are symmetrical, however, the atoms pull equally on the electrons and the charge distribution is uniform.Do we breathe out carbon dioxide?
When we exhale, we breathe out less oxygen but more carbon dioxide than we inhale. The carbon we breathe out as carbon dioxide comes from the carbon in the food we eat. The carbon dioxide is dissolved in the blood, carried to the lungs by the circulation, and breathed out.Is oxygen a molecule?
Oxygen in the atmosphere is a molecule because it contains molecular bonds. It is not a compound because it is made from atoms of only one element - oxygen. This type of molecule is called a diatomic molecule, a molecule made from two atoms of the same type.