What is electrode filling solution?
What is electrode filling solution?
Mettler Toledo™ KCl 3M Electrode Filling Solution is an electrolyte solution that enables optimum performance of pH and ion-selective electrodes. This bridge electrolyte contains no silver ions than can contaminate samples.
What is a Ross electrode?
For more than 40 years, ROSS pH electrodes have been helping to provide accuracy, stability and rapid response. ROSS pH electrodes are available in a variety of styles such as the clog free Sure-Flow junction, glass or epoxy electrodes, flat surface, semi-micro, micro, and spear tip designs.
What fluid is used to fill a liquid filled electrode?
The slope of the electrode increases linearly with temperature. The potential inside the pH glass bulb is fixed by the filling solution, and the reference electrode potential is constant.
How do you make a pH electrode filling solution?
2. Store your electrode in a solution of 4M kCl solution, If 4M KCl is not available, use a pH 4 Buffer We use 4M KCL + pH 4 buffer (Volume ration 1:1) it is better. DO NOT store electrode in distilled or deionized water, this will cause ions to leach out of the glass bulb and render your electrode useless. 3.
How do you fix a pH electrode?
Reconditioning Electrodes
- Method 1: Soak the electrode in a 0.4 M of HCl (hydrochloric acid) for 10 minutes, then rinse the electrode with deionized or distilled water.
- Method 2: Soak the electrode in a 3.8 or 4.0 M KCl (potassium chloride) solution heated to 50oC for one hour.
How do you refill a pH meter?
Remove the filling hole plug, and fill the pH electrode with electrolyte to just below the refill hole. Leave a small amount of space below the refill hole to help minimize electrolyte leakage and build up of KCl crystallization at the filling hole.
How do you make 3 molar KCl?
To make 125 mL of 3M KCl
- Using digital scale, place 27.96 g KCl powder into weigh boat.
- In 150 mL flask, mix 27.96 g KCl with 100 mL H2O.
- Put flask on hotplate/stirrer with stirbar until dissolved.
- Add H2O to bring volume to 125 mL.
What is a gel filled pH electrode?
The sension Gel-filled pH Electrode responds to the hydrogen ion concentration (activity) by developing an electrical potential. At a constant temperature, this potential varies linearly with the pH of the solution being measured.
Is it proper to store the electrode in deionized water?
DO NOT store electrode in distilled or deionized water—this will cause ions to leach out of the glass bulb and render your electrode useless. After storage, you may notice white KCl crystals forming outside your electrode.
How long do pH electrodes last?
When using and storing a pH electrode the usual way, the expected lifetime is 1 to 3 years. Some customers use their electrodes for up to 8 years! However there are a number of factors that can contribute to shortening the lifetime of an electrode. One of these is the use of hot and very alkaline samples.
How do you recharge a pH electrode?
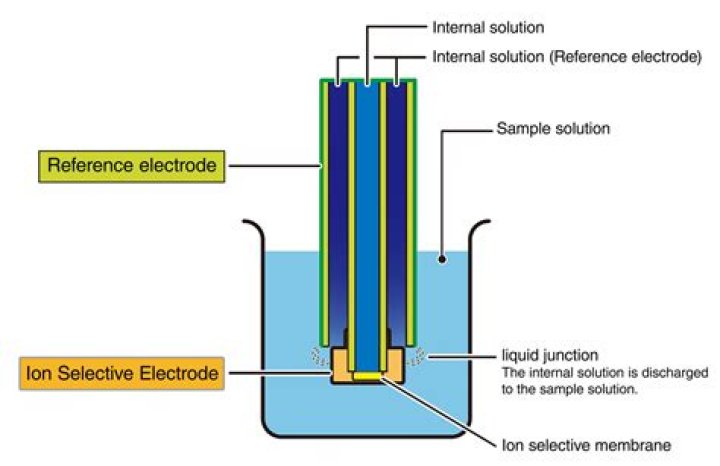
How do you fill a reference electrode?
Filling Solution Both reference electrodes are filled with Saturated KCl. To fill the reference electrode, slide the rubber grommet located at the top of the electrode down. This will reveal a small filling port where saturated KCl can be added.