What is the atomic number of oxygen?
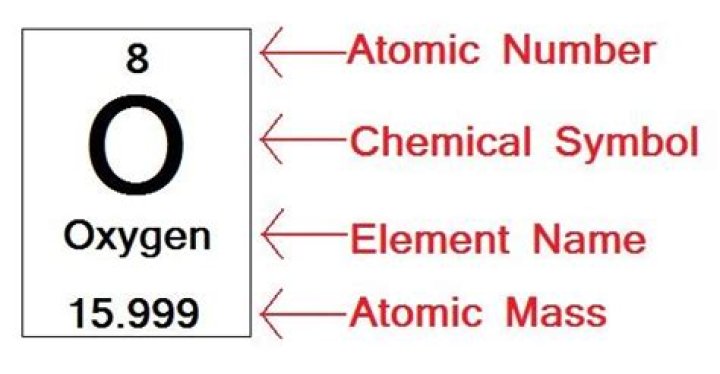
What is the atomic number of oxygen?
8
Oxygen/Atomic number
How many neutrons does nickel 63?
List of isotopes
| Nuclide | Z | N |
|---|---|---|
| Excitation energy | ||
| 61 Ni | 28 | 33 |
| 62 Ni | 28 | 34 |
| 63 Ni | 28 | 35 |
What is the same about oxygen 16 17 and 18?
Isotopes are elements that contain the same amount of protons, but differ in the number of neutrons in their nuclei. Therefore, oxygen 16 has 8 protons and 8 neutrons, oxygen 17 has 8 protons and 9 neutrons, and oxygen 18 has 8 protons and 10 neutrons.
What is the atomic number for oxygen 16?
Oxygen-16 (16O) is a stable isotope of oxygen, having 8 neutrons and 8 protons in its nucleus. It has a mass of 15.99491461956 u. Oxygen-16 is the most abundant isotope of oxygen and accounts for 99.762% of oxygen’s natural abundance….Oxygen-16.
| General | |
|---|---|
| Symbol | 16O |
| Names | oxygen-16, O-16, Oxygen – 16 |
| Protons | 8 |
| Neutrons | 8 |
What is atomic weight of oxygen?
15.999 u
Oxygen/Atomic mass
Another example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows: Atomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) 0.99759(15.995u)+0.00037(16.995u)+0.00204(17.999u)=15.999u.
What is the atomic structure of oxygen?
The nucleus of oxygen consists of 8 protons (purple) and 8 neutrons (green). It features eight electrons (yellow) placed on the electron shells (rings).
What is the mass of nickel-63?
62.929669
Nickel-63
| PubChem CID | 104905 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | Ni |
| Synonyms | Nickel-63 UNII-C4L55RH4J6 C4L55RH4J6 13981-37-8 Nickel, isotope of mass 63 More… |
| Molecular Weight | 62.929669 |
How many protons and neutrons does nickel-63 have?
All nickel atoms contain 28 protons (Z=28) and various numbers of neutrons (typically N=28 to 38 neutrons) within the atom’s nucleus.
What is the atomic number of oxygen-18?
CHEBI:33815 – oxygen-18 atom
| ChEBI Name | oxygen-18 atom |
|---|---|
| ChEBI ID | CHEBI:33815 |
| Definition | The stable isotope of oxygen with relative atomic mass 17.999160 and 0.205 atom percent natural abundance. |
| Stars | This entity has been manually annotated by the ChEBI Team. |
| Supplier Information | No supplier information found for this compound. |
What is the atomic number of uranium 235?
92
Fact box
| Group | Actinides | 1135°C, 2075°F, 1408 K |
|---|---|---|
| Period | 7 | 4131°C, 7468°F, 4404 K |
| Block | f | 19.1 |
| Atomic number | 92 | 238.029 |
| State at 20°C | Solid | 234U, 235U, 238U |
What is the atomic number of oxygen 18?
What is the atomic number of oxygen-17?
Properties of Oxygen-17 Isotope:
| Properties of Oxygen-17 Isotope: | OXYGEN-17 |
|---|---|
| Neutron Number (N) | 9 |
| Atomic Number (Z) | 8 |
| Mass Number (A) | 17 |
| Nucleon Number (A) | 17 |