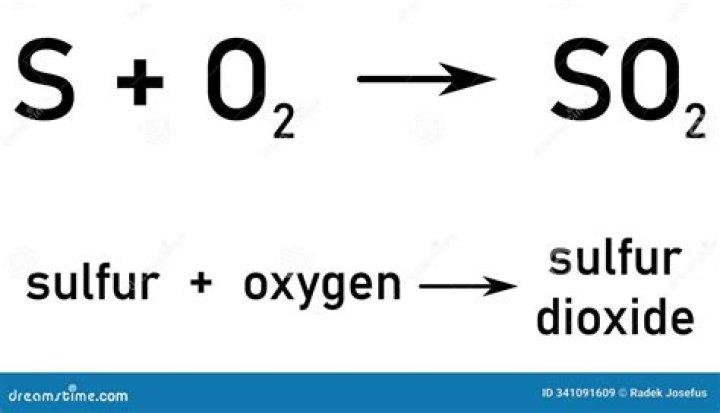The compound formed between sulfur and barium is barium sulfide BaS..
Considering this, what is the correct formula for the compound that forms between barium and sulfur?
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Barium sulfide is the inorganic compound with the formula BaS.
Likewise, what is the chemical formula for the compound formed between strontium and sulfur? Strontium sulfide is the inorganic compound with the formula SrS. It is a white solid. The compound is an intermediate in the conversion of strontium sulfate, the main strontium ore called celestite, to other more useful compounds.
Just so, what is the chemical formula for the compound formed between barium and iodine?
The chemical formula of barium and iodine compound is BaI² (I= i)
What is the chemical formula for the compound formed between calcium and iodine?
Calcium iodide (chemical formula CaI2) is the ionic compound of calcium and iodine.
Related Question Answers
What is the formula for ba2+ and F?
The formula for the barium ion is Ba2+ . Fluorine is a group 17 element.What is the formula for rubidium nitride?
Rubidium Nitride Rb3N Molecular Weight -- EndMemo.What is a molecular compound?
A molecular compound can be defined as a compound where the atoms share electrons through covalent bonds. It is also known as a covalent compound. The covalent bonds are what hold the molecule together. When atoms share electrons, they are able to have a full outer electron shell.What is the name of the compound NaBr?
Sodium Bromide
What is the formula for potassium and sulfur?
K2S
What is the formula for the compound formed from calcium and oxygen?
The formula for the compound formed from calcium and oxygen is CaO, which is calcium oxide. Calcium and oxygen form an ionic bond due to the number ofIs baf2 a molecular compound?
Barium fluoride (BaF2) is an inorganic compound with the formula BaF2. It is a colorless solid that occurs in nature as the rare mineral frankdicksonite. It adopts the fluorite structure and at high pressure the PbCl2 structure.Is barium a metal?
Barium is a chemical element with the symbol Ba and atomic number 56. It is the fifth element in group 2 and is a soft, silvery alkaline earth metal. Because of its high chemical reactivity, barium is never found in nature as a free element.What is the name of Cos?
Carbonyl sulfide is the chemical compound with the linear formula OCS. Normally written as COS as a chemical formula that does not imply its structure, it is a colourless flammable gas with an unpleasant odor. It is a linear molecule consisting of a carbonyl group double bonded to a sulfur atom.What is the name of kch3co2?
Potassium acetate. From Wikipedia, the free encyclopedia. Potassium acetate. Names.What type of compound is na3p?
Sodium phosphide is the inorganic compound with the formula Na3P. It is a black solid.Is BaBr soluble?
Barium bromide has the molecular formula of BaBr2. Like barium chloride, it is soluble in water but is toxic if ingested. In aqueous solution, BaBr2 behaves as a simple salt.What is the correct name for BaI2?
Barium iodide
What is the chemical formula for the compound formed between lithium and bromine?
Lithium bromide (LiBr) is a chemical compound of lithium and bromine. Its extreme hygroscopic character makes LiBr useful as a desiccant in certain air conditioning systems.What color is bai2?
Barium iodide is an inorganic compound with the formula BaI2. The compound exists as an anhydrous and a hydrate (BaI2(H2O)2), both of which are white solids.Is bai2 soluble in water?
Properties of Barium Iodide (anhydrous) Appearance: it is a white or off-white crystalline powder. Solubility: This inorganic compound is soluble in water. It is also soluble in ethanol and acetone.What is the Colour of barium sulphate?
white
What is Sr3P2?
Sr3P2 molecular weight This compound is also known as Strontium Phosphide.How do you find the empirical formula?
What is the empirical formula of the compound? Start with the number of grams of each element, given in the problem. Convert the mass of each element to moles using the molar mass from the periodic table. Divide each mole value by the smallest number of moles calculated.