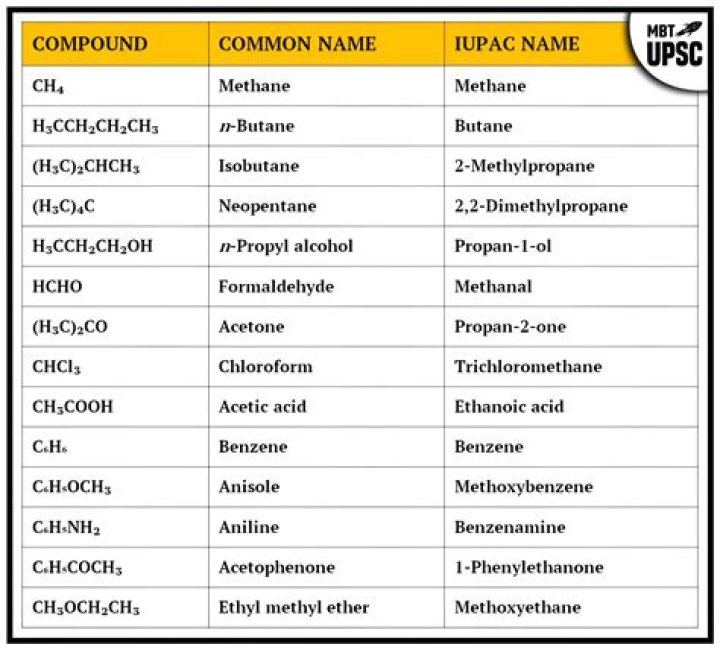
Octane is an alkane with the chemical formula C8H18. It has 18 isomers. Octane is a straight chain alkane composed of 8 carbon atoms. It has a role as a xenobiotic..
Likewise, people ask, what is octane used for?
It just prevents the air-fuel mixture from igniting before the spark plug does it. Firing the air-fuel mixture at the proper time gives you the maximum power your engine was designed to get. Using higher-octane gasoline than your engine is designed to utilize is only wasting your money.
Additionally, is octane acidic or basic? Petrol, which is mostly octane, doesn't dissolve in water (to any appreciable amount) and it doesn't react with water either, so therefore it can't be considered either an acid or base.
Similarly, what is octane formula?
C8H18
What are the 18 isomers of octane?
18 Isomers of Octane - C8H18
- Octane.
- 2-Methylheptane.
- 3-Methylheptane.
- 4-Methylheptane.
- 2,2-Dimethylhexane.
- 2,3-Dimethylhexane.
- 2,4-Dimethylhexane.
- 2,5-Dimethylhexane.
Related Question Answers
Does octane increase horsepower?
Octane does not offer any better fuel mileage, increase engine horsepower, or make the engine start quicker. Higher octane only reduces the likelihood of engine knock or ping. Because higher octane gas burns slower, it is more resistant to knock when subjected to higher RPM and cylinder pressures.Does octane combust?
2 C8H18 + 25 O2 ---> 16 CO2 +18 H2O. Combustion engines work with gasoline based on stoichiometric principle or fuel to air ratio. This is because lower octane gas ignites at lower temperatures and is as such more flammable. Higher octane gas requires more compression and higher temperatures to burn efficiently.Does octane affect gas mileage?
Car fuel mileage is not affected by a higher octane level. Many believe that the higher the octane level has a direct benefit on your fuel economy. Not only will the octane level not effect your gas mileage, but it does not even increase the amount of power your car puts out.What drug does octane use?
FDA laboratory analysis confirmed that Fuel Up High Octane contains hydroxythiohomosildenafil. Hydroxythiohomosildenafil is structurally similar to sildenafil, the active ingredient in the FDA-approved prescription drug Viagra, used to treat erectile dysfunction (ED).What does octane do for your engine?
The octane rating of gasoline tells you how much the fuel can be compressed before it spontaneously ignites. When gas ignites by compression rather than because of the spark from the spark plug, it causes knocking in the engine.Does octane evaporate?
Octane does not evaporate. Octane is a rating not a chemical. What DOES evaporate first in gasoline is the "light ends" or light aromatics, these are the v.o.c.'s that make your engine easy to start.Where is octane found?
Octane is a hydrocarbon and most hydrocarbon groups are found in petroleum. Petroleum is an oil which is found in nature and under the surface of the earth. It is a mixture of crude oil and natural gas. Crude oil and natural gas are found in the nature, such as under the ground and the ocean.What does putting higher octane gas do?
The higher the octane, the greater its ability to prevent the unruly type of combustion engineers call detonation. The goal is to ignite the fuel mixture solely with the spark plug, rather than from the heat in the cylinder, to prevent detonation, which can seriously damage high-performance engines.What is the chemical formula for gasoline?
The bulk of a typical gasoline consists of a homogeneous mixture of small, relatively lightweight hydrocarbons with between 4 and 12 carbon atoms per molecule (commonly referred to as C4–C12). It is a mixture of paraffins (alkanes), olefins (alkenes) and cycloalkanes (naphthenes).Who discovered octane?
Edwin L. Drake
What does N octane mean?
Modern octane ratings of gasoline are given octane ratings equal to those from this original heptane/octane scale. For example, n-octane (the straight chain of 8 carbon atoms with no branching) has a -10 (negative) octane rating, while pure 2,2,4-trimethylpentane (a highly branched octane) has an octane rating of 100.Is Octane an alcohol?
Both alcohols have a high octane rating, with ethanol at 109 RON (Research Octane Number), 90 MON (Motor Octane Number), (which equates to 99.5 AKI) and methanol at 109 RON, 89 MON (which equates to 99 AKI). Ethanol is, and has always been, a superior fuel in all ways.What is octane in science?
Octane is an organic chemical, specifically an alkane, with formula C8H18. (8 carbon alkene). Octane ratings are ratings used to represent the anti-knock performance of petrol by giving the percentage of octane in an octane-heptane mixture that would have the same performance.Is Octane a fossil fuel?
Most cars use unleaded high octane gasoline as fuel. High octane gas is a mixture of at least 500 various hydrocarbons ranging from 5 to 12 carbon atoms. Crude oil, also known as petroleum, is a type of fossil fuel. There are different grades of crude oil and each grade is used for different purposes.Is Octane a compound?
Octane is a compound because on reaction with oxygen, it forms carbon dioxide and water. The source of carbon and hydrogen both must be octane, thus octane is a compound.What is the octane number of fuel?
Octane rating is the measure of a fuel's ability to resist "knocking" or "pinging" during combustion, caused by the air/fuel mixture detonating prematurely in the engine. In the U.S., unleaded gasoline typically has octane ratings of 87 (regular), 88–90 (midgrade), and 91–94 (premium).How dangerous is heptane?
Health risks Acute exposure to heptane vapors can cause dizziness, stupor, incoordination, loss of appetite, nausea, dermatitis, chemical pneumonitis, or unconsciousness, possible peripheral neuropathy.Does octane dissolve in water?
Since we've established that octane is considered to be non-polar, it will not be soluble in water, since water is a polar solvent. This will happen because octane (hydrocarbons in general) contains neither ionic groups, nor polar functional groups that can interact with water molecules.What is c3h18?
The hydrocarbon octane, C3H18, has eight carbons per. molecule and is a component of gasoline, which is a mixture of many different. sorts of hydrocarbons. The hydrocarbon polyethylene contains hundreds of. carbon and hydrogen atoms per molecule.