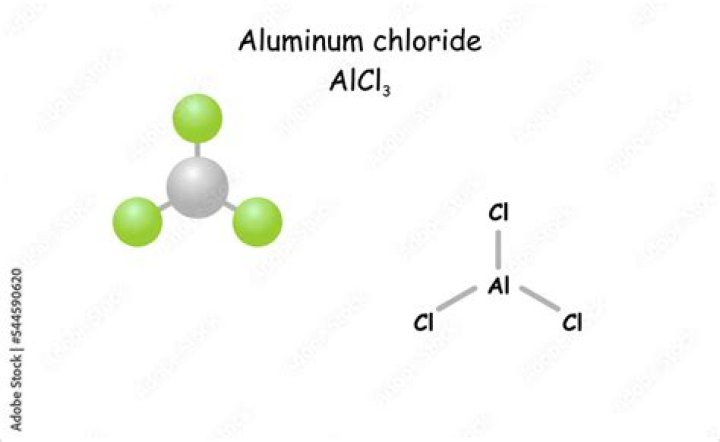
What is the empirical formula for aluminum chloride?
.
Similarly, it is asked, what is the chemical equation for Aluminium chloride?
AlCl3
Also Know, what is the charge of aluminum chloride? Explanation: Al has a charge of +3. Cl has a charge of -1. Hence 3 chloride ions are required to neutralize charge of +3 on aluminium and the compound formed is AlCl3 .
Also asked, how do you write aluminum chloride?
Aluminum chloride is a chemical compound with the chemical formula AlCl3.
How do you balance aluminum chloride?
In order to balance Al + Cl2 → AlCl3 you'll need to be sure to count all of Aland Cl atoms on each side of the chemical equation. Once you know how many of each type of atom you have you can only change the coefficients (the numbers in front of atoms or compounds) in order to balance the equation.
Related Question AnswersWhat state of matter is aluminum chloride?
Aluminium chloride (AlCl3), also known as aluminium trichloride, is the main compound of aluminium and chlorine. It is white, but samples are often contaminated with iron(III) chloride, giving it a yellow color. The solid has a low melting and boiling point.What is the structure of Aluminium chloride?
Structure of Aluminium Chloride (AlCl3) When AlCl3 is in solid-state it features a cubic close-packed layered structure. In this case, its coordination geometry will be octahedral. When aluminium chloride is in a liquid or molten state it exists as a dimer. Here its coordination geometry will be tetrahedral.Is aluminum chloride safe to use?
Aluminum chloride is a definite life saver in the sweating department, but there are some side effects. When it comes into contact with skin it can cause burning, stinging, itching, and tingling. Not to worry, though; these topical irritations occur with pure aluminum chloride.How many ions are in aluminum chloride?
The chemical formula of aluminum chloride is AlCl3. Let's see why three chloride ions are needed for one aluminum ion. A general rule in chemistry is that an atom will gain or lose electrons to have a total of eight electrons in the outermost shell of its electron cloud.Is AlCl3 an acid or base?
AlCl3 is acidic. But wait, now I've an acid, i.e. hydrochloric acid (HCl) and probably a base (Al(OH)3). Close enough, but Al(OH)3 is actually amphoteric in nature, which means it can behave both as an acid and a base.What is aluminum chloride used for?
Aluminum chloride hexahydrate is an antiperspirant that works by affecting the cells that produce sweat. Aluminum chloride hexahydrate topical (for the skin) is used to treat excessive sweating, also called hyperhidrosis.Is aluminum chloride a gas?
Anhydrous aluminum chloride (aluminum trichloride, AlCl3) is an odorless, white or yellow crystalline solid that reacts violently with water to liberate hydrogen chloride (HCL) gas.What antiperspirants have aluminum chloride?
Popular over the counter antiperspirants for hyperhidrosis| Brand | Forms | Compount |
|---|---|---|
| Driclor | Roll-on, Solution | aluminum chloride. |
| SweatBlock | Wipes | aluminum chloride. |
| Dove | Solid | aluminum zirconium tetrachlorohydrex gly (20%) |
| Secret Clinical Strength | Clear Gel | Aluminum zirconium tetrachlorohydrex Gly |
What is the subscript for aluminum?
Since the charges are not equal in magnitude, the charge on the aluminum ion (+3) becomes the subscript for the sulfate ion. The charge on the sulfate ion (-2) becomes the subscript for the aluminum ion.Does aluminum chloride dissolve in water?
WaterHow do you make anhydrous Aluminium chloride?
Usually from 190 to 200 g of aluminum chloride may be obtained from 50 g of aluminum. A solution of aluminum chloride can be prepared directly by the action of hydrochloric acid on the aluminum, but if this solution is evaporated to dryness, the solid that is left is the aluminum oxide instead of the aluminum chloride.Is aluminum chloride a binary compound?
Can you name binary ionic compounds?Binary Ionic Compounds.
| A | B |
|---|---|
| aluminum nitride | AlN |
| aluminum phosphide | AlP |
| magnesium fluoride | MgF2 |
| magnesium chloride | MgCl2 |