What is the importance of ozone layer?
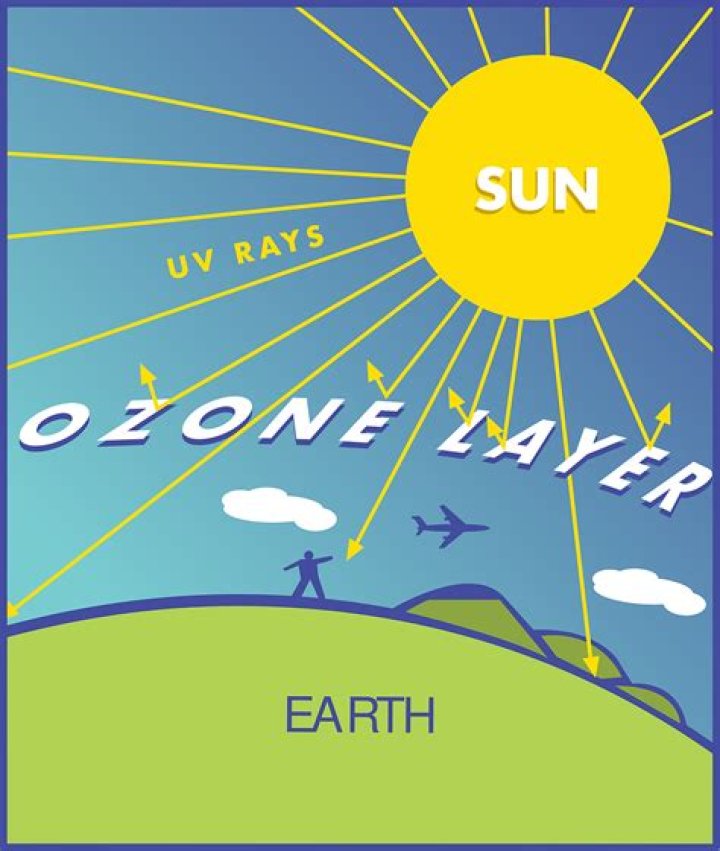
What is the importance of ozone layer?
Why is Ozone Layer important? Ozone protects the Earth from harmful ultraviolet (UV) rays from the Sun. Without the Ozone layer in the atmosphere, life on Earth would be very difficult. Plants cannot live and grow in heavy ultraviolet radiation, nor can the planktons that serve as food for most of the ocean life.
What is the purpose of resonance structures?
Resonance structures are used when one Lewis structure for a single molecule cannot fully describe the bonding that takes place between neighboring atoms relative to the empirical data for the actual bond lengths between those atoms.
What are the uses of ozone?
Ozone and it’s application
- Water purification: drinking water, bottled water, swimming pools, industrial wastewaters cooling towers, groundwater remediation, wastewater re-use.
- Air purification: gas emissions from industry, conditioning system.
- Pulp bleaching for chlorine-free production of paper.
- Organic synthesis.
What statement best describes an O3 molecule?
An ozone molecule contains one single bond and one double bond.
How is ozone layer formed state its importance to all life forms on Earth?
Answer: Ozone is formed when heat and sunlight cause chemical reactions between oxides of nitrogen (NOX ) and Volatile Organic Compounds (VOC), which are also known as Hydrocarbons. This reaction can occur both near the ground and high in the atmosphere.
What is the resonance structure of O3?
Is ozone a resonance structure? Ozone, or O3, has two major structures of resonance that contribute equally to the molecule’s overall hybrid structure. All structures reflect the 18 valence electrons required-6 out of 3 bonds and 12 as lone pairs placed on the oxygen atoms.
What are the concepts of resonance explain?
Resonance is a way of describing delocalized electrons within certain molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. A molecule or ion with such delocalized electrons is represented by several resonance structures.
What is structure of ozone?
The structure of ozone has 3 oxygen atoms, but steric hindrance prevents it from forming a triangular structure, with each O atom forming the expected 2 bonds. Instead each Oxygen forms only 1 bond, with the remaining negative charge being spread throughout the molecule.
What is properties of ozone?
Ozone is a bluish gas with a boiling point at -112oC. At atmospheric pressure, ozone can partially dissolve in water. At standard pressure and temperature, the solubility of ozone is thirteen times that of oxygen. The oxidation potential of 2.07 volts proves that ozone is a strong oxidizer.
What is the bond order of the bonds between O atoms in the ozone molecule o3?
1.5
From the Lewis structure, we see that the bond order for O2 is 2 (a double bond), whereas the bond order for O3 is 1.5 (one and a half bonds).
Which resonance structure is likely to contribute most to the correct structure of N2O?
Structure B contributes the most to the correct overall structure of N2O because it minimizes the number of formal charges and the negative formal charge resides on oxygen, which is more electronegative than nitrogen.
What is ozone how it is formed state the significance of the ozone layer?
Ozone layer is located at the higher levels of the atmosphere, Le., in stratosphere. → Ozone is formed by the action of DV (Ultra- violet) radiation on oxygen (O2) molecule. → The higher energy of DV radiation convert some oxygen (O2) molecules into free oxygen (O) atoms.



