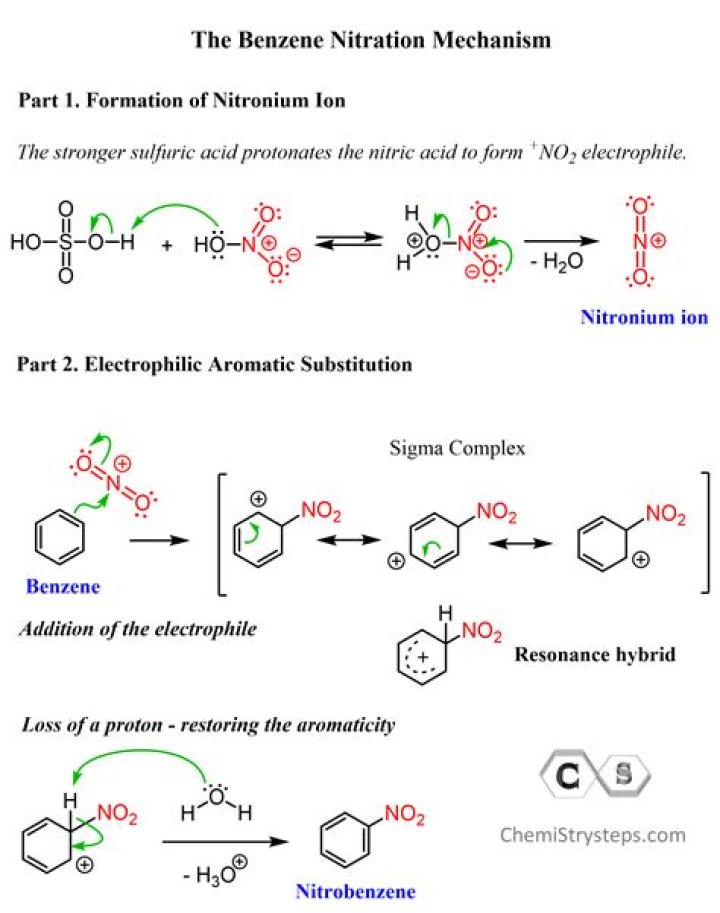
The mechanism for nitration of benzene: Step 1: Nitric acid accepts a proton from sulphuric acid and then dissociates to form nitronium ion. Step 2: The nitronium ion acts as an electrophile in the process which further reacts with benzene to form an arenium ion..
Also to know is, what do you understand by nitration explain the mechanism of nitration of benzene?
Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitro group, NO2. Benzene is treated with a mixture of concentrated nitric acid and concentrated sulphuric acid at a temperature not exceeding 50°C. The mixture is held at this temperature for about half an hour.
Subsequently, question is, what is the electrophile in the nitration of benzene? The electrophile is the "nitronium ion" or the "nitryl cation", NO+2. This is formed by reaction between the nitric acid and the sulphuric acid.
Just so, what is nitration and its mechanism?
In general, nitration is the process of adding a nitro group to an organic substrate. The most common form of nitration is adding the nitro group to an aromatic ring system. The nitration reaction mechanism involves: Formation of the nitronium ion by the action of nitric and sulfuric acids.
What is the role of h2so4 in nitration of benzene?
The source of the nitronium ion is through the protonation of nitric acid by sulfuric acid, which causes the loss of a water molecule and formation of a nitronium ion. The first step in the nitration of benzene is to activate HNO3 with sulfuric acid to produce a stronger electrophile, the nitronium ion.
Related Question Answers
What is nitration of benzene used for?
Nitration is used to add nitrogen to a benzene ring, which can be used further in substitution reactions. The nitro group acts as a ring deactivator. Having nitrogen present in a ring is very useful because it can be used as a directing group as well as a masked amino group.What conditions are used for nitration?
Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitro group, NO2. Benzene is treated with a mixture of concentrated nitric acid and concentrated sulfuric acid at a temperature not exceeding 50°C. The mixture is held at this temperature for about half an hour.What are the reactions of benzene?
Benzene reacts with chlorine or bromine in an electrophilic substitution reaction, but only in the presence of a catalyst. The catalyst is either aluminum chloride (or aluminum bromide if you are reacting benzene with bromine) or iron.Why benzene does not react with bromine?
Reactivity of Cyclohexene and Benzene However when bromine is added to benzene, the bromine remains orange and there is no reaction. This is because: Benzene has delocalised electrons spread over 6 carbon atoms, whereas alkenes have localised electrons above and below the 2 carbon atoms in the double bond.What is nitration reaction?
Nitration is a general class of a chemical process for the introduction of a nitro group into an organic chemical compound. Nitration reactions are notably used for the production of explosives, for example the conversion of guanidine to nitroguanidine and the conversion of toluene to trinitrotoluene.What is electrophilic reagent?
Electrophilic reagents are chemical species which, in the course of chemical reactions, acquire electrons, or a share in electrons, from other molecules or ions.What is a nitrating agent?
NITRATING ACID, MIXTURE, (WITH <= 50% NITRIC ACID) is a mixture of nitric with sulfuric acids. Reacts as a strong acid. Exothermically neutralizes all kinds of bases (including amines and amides). Acts as an oxidizing agent and a nitrating agent.What does acylation mean?
In chemistry, acylation (or alkanoylation) is the process of adding an acyl group to a compound. The compound providing the acyl group is called the acylating agent. Acylation can be used to prevent rearrangement reactions that would normally occur in alkylation.What is sulfonation reaction?
From Wikipedia, the free encyclopedia. Aromatic sulfonation is an organic reaction in which a hydrogen atom on an arene is replaced by a sulfonic acid functional group in an electrophilic aromatic substitution. Aryl sulfonic acids are used as detergents, dye, and drugs.What is halogenation reaction?
Halogenation is a reaction that occurs when one or more halogens are added to a substance. Halogens comprise the seventh column in the periodic table and include fluorine, chlorine, bromine, iodine, and astatine. The resulting product of a halogenation reaction is known as a halogenated compound.What is bromination reaction?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution. Bromination of a benzylic position by a free radical substitution reaction.Why nitration of toluene is easier than benzene?
Organic Chemistry: Why nitration of toluene is easier than benzene? The CH3 group attached to the benzene ring increases the electron density through + H effect. The electrophile attacks to the substrate having high electron density. Electrophilic aromatic substitution reaction is a second order reaction.How do you get Ethyne from benzene?
Benzene is prepared from ethyne by the process of cyclic polymerization. In this process, Ethyne is passed through a red hot iron tube at 873 K. The ethyne molecule then undergoes cyclic polymerization to form benzene.Are nitro groups Basic?
The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. The nitro group is also strongly electron-withdrawing. Because of this property, C−H bonds alpha (adjacent) to the nitro group can be acidic.What is the rate determining step in the nitration of benzene?
Nitration of Benzene The first step involves the generation of electrophile NO2+ which is formed from the reaction between conc HNO3 and conc H2SO4. In the second step, which is the rate determining step or slow step, benzene will react with the electrophile NO2+.Why is benzene iodination so hard?
Why is Iodination of Benzene Difficult? For satisfying this condition, electron donating groups attached to the phenyl ring making it more nucleophilic are preferred over unsubstituted Benzene. Also, the electrophilicity of the halogen is increased by using a Lewis acid catalyst thereby making it more reactive.Is nh4+ A Nucleophile?
Let us look at the structure of the ammonium ion (NH4+). In an ammonium ion, nitrogen is bonded to hydrogen atoms and shares electrons with them. This causes all the orbitals to be fully filled and hence, nitrogen does not have space for any additional electrons. Thus, the ammonium ion is not an electrophile.Is no2 an electrophile?
Hence an unpaired electron remains on the nitrogen atom which is then ionised to get a +ve charge (formal charge) on nitrogen.As nitrogen doesnt have an octet around it, hence it always craves for an electron and also the resonance between the N-O bonds make it a strong electrophile.How do you alkylate benzene?
Alkylation means substituting an alkyl group into something - in this case into a benzene ring. A hydrogen on the ring is replaced by a group like methyl or ethyl and so on. Benzene is treated with a chloroalkane (for example, chloromethane or chloroethane) in the presence of aluminium chloride as a catalyst.