What is the molar mass of fluorine?
18.998403 u
.
Correspondingly, what is the molar mass of fluorine gas?
37.99681 g/mol
how much is fluorine a gram? fluorine costs about 190 dollars per gram. It is only present in a few ores, and in its pure form is a highly poisonous gas, making the refinement process more difficult. Fluorine cost about $190 per 100 grams. Fluorine is the chemical element in the periodic table that has the symbol F and atomic number nine.
Likewise, how many moles are in fluorine?
The molar mass (M) of fluorine is 19.0 g/mol. Therefore, the number of moles (n) for fluorine is: [n = 200 grams ÷ 19.0 g/mol] = 10.5 moles. There are 10.5 moles in 200 grams of fluorine.
What is the formula mass of f2?
37.99681 g/mol
Related Question AnswersHow do you determine molar mass?
Key Points- The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol).
- The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms.
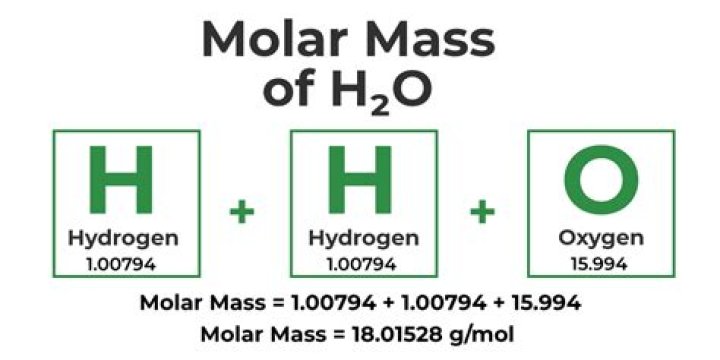
How many moles are there in 54 g of water?
Explanation: There are 6.022×1023 molecules in a mole. There are 18.01528 grams of water per mole of water. These can be figured out using the concept of moles and molecular weight.What is the mass in grams of 0.123 moles of fluorine gas?
We assume you are converting between moles Fluorine and gram. You can view more details on each measurement unit: molecular weight of Fluorine or grams The molecular formula for Fluorine is F. The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles Fluorine, or 18.9984032 grams.What is the formula for fluorine?
The chemical formula for fluorine gas is F2. The electronic configuration of the fluorine atom is 1s2 2s2 2p5.Where is fluorine found?
Fluorine occurs naturally in the earth's crust where it can be found in rocks, coal and clay. Fluorides are released into the air in wind-blown soil. Fluorine is the 13th most aboundant element in the Earth's crust: 950 ppm are contanined in it.What is atomic mass in science?
Atomic mass or weight is the average mass of the protons, neutrons, and electrons in an element's atoms.What is fluorine used for?
Uses of Fluorine Compounds of fluorine, including sodium fluoride, are used in toothpaste and in drinking water to prevent dental cavities. Hydrofluoric acid can dissolve glass and is used to etch the glass in light bulbs and in other products.How do we find atomic mass?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. Example: Find the atomic mass of an isotope of carbon that has 7 neutrons. You can see from the periodic table that carbon has an atomic number of 6, which is its number of protons.What is the mass number of calcium?
Answer and Explanation: The mass number of calcium is 40amu. The mass number is equal to the number of protons added to the number of neutrons in an atom. Each subatomicWhat is the molar mass of cl2?
The molar mass of Cl2 is 70.906. To calculate molar mass of a chemical compound enter its formula and click 'Calculate! '.How many particles are there in one mole?
In science, we have a name for this, called Avogadro's number, and it describes the number of representative particles in one mole of a substance. The inverse mole unit tells us there are 6.022×1023 particles of something *per mole*.Is carbon a diatomic molecule?
Carbon is not a diatomic element. Diatomic elements are those where two atoms of the element can join to form a single molecule.How do you convert from moles to grams?
Moles to Grams Conversion Formula. In order to convert the moles of a substance to grams, you will need to multiply the mole value of the substance by its molar mass. More commonly written for this application as: where, is the molar mass of the substance.What is NaF chemistry?
Sodium fluoride is an inorganic salt, which is an important source of the fluoride ion for many applications. Formula and structure: The chemical formula of sodium fluoride is NaF and its molar mass is 41.99 g/mol. It is a simple ionic compound, made of the sodium (Na+) cation and fluoride (F-) anion.What is a mole in chemistry?
The mole is the unit of amount in chemistry. A mole of a substance is defined as: The mass of substance containing the same number of fundamental units as there are atoms in exactly 12.000 g of 12C. Fundamental units may be atoms, molecules, or formula units, depending on the substance concerned.How many moles are in f2?
The answer is 37.9968064. We assume you are converting between grams F2 and mole. You can view more details on each measurement unit: molecular weight of F2 or mol The SI base unit for amount of substance is the mole. 1 grams F2 is equal to 0.026318001293919 mole.How many moles are in 15 grams of lithium?
1 Answer. Approx 52 moles.How do you convert molecules to moles?
Converting between molecules and moles is done by either multiplying by or dividing by Avogadro's number:- To go from moles to molecules, multiply the number of moles by 6.02 x 1023.
- To go from molecules to moles, divide the numbers of molecules by 6.02 x 1023.



