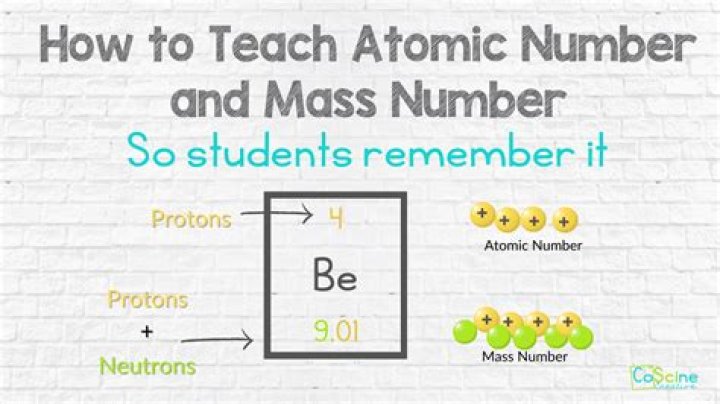
54
.
Subsequently, one may also ask, what is the normal phase of xenon?
| Name | Xenon |
| Boiling Point | -108.12° C |
| Density | .005887 grams per cubic centimeter |
| Normal Phase | Gas |
| Family | Noble Gases |
Subsequently, question is, what element has 54 electrons and a mass number of 132? Xenon
Beside above, what's the number of neutrons in xenon?
54
How many protons neutrons and electrons does XE 133 possess?
Description: Xenon Xe-133 is a radioisotope of xenon, an element with 54 protons, having gamma emissions and a physical half-life of 5.27 days.
Related Question Answers
Does xenon react with anything?
Xenon reacts directly with fluorine because fluorine is a very powerful oxidizing agent (hence it gets reduced and it gains an electron from Xenon). Xenon does not react directly with oxygen. Xenon reacts with water molecules in order to form xenon oxides.Is Xenon heavier than air?
Xenon (Xe), chemical element, a heavy and extremely rare gas of Group 18 (noble gases) of the periodic table. More than 4.5 times heavier than air, xenon is colourless, odourless, and tasteless.What is the cost of xenon?
Xenon currently costs approximately US $10.00 per liter.Why is Xenon so unreactive?
Chemists originally believed that the noble gases could not form compounds , because their full valence shell of electrons made them chemically stable and unreactive. The inner electrons shield the outermost electrons, so they are less strongly attracted to the nucleus.What is xenon used for today?
Uses of Xenon Xenon is used in photographic flashes, in high pressure arc lamps for motion picture projection, and in high pressure arc lamps to produce ultraviolet light. It is used in instruments for radiation detection, e.g., neutron and X-ray counters and bubble chambers.Where is xenon most commonly found?
Xenon is a trace gas found in the Earth's atmosphere to the extent of about one part in 20 million, According to the Los Alamos National Laboratory. This makes it very rare. It is also found in Mars' atmosphere at 0.08 ppm. This noble gas can also be found down on Earth.What creates xenon 129?
Xe is produced by beta decay of 129I (half-life: 16 million years); 131mXe, 133Xe, 133mXe, and 135Xe are some of the fission products of both 235U and 239Pu, so are used as indicators of nuclear explosions. Xenon isotopes are also a powerful tool for understanding terrestrial differentiation.Why is xenon used in lights?
The earth's atmosphere is about 0.0000087% xenon. Xenon produces a brilliant white flash of light when it is excited electrically and is widely used in strobe lights. The light emitted from xenon lamps is also used to kill bacteria and to power ruby lasers.How do you figure out neutrons?
Take note that the nucleus of an atom is composed of protons and neutrons. And the number of particles present in the nucleus is referred as mass number (Also, called as atomic mass). So, to determine the number of neutrons in atom, we only have to subtract the number of protons from the mass number.What element has 16 neutrons?
phosphorus
How many neutrons does gold have?
118 neutrons
What does xenon look like?
Its atomic number is 54 and it is a member of the noble gases, which are found in Group 18 on the periodic table. Xenon is odorless, colorless, and heavier than the air found in our atmosphere. Because of its ability to emit light when electricity is added, it can be used in specialized lamps.How many neutrons are in lead?
Since lead has 82 protons, the number of neutrons in an atom of lead-204 is 204-82=122. Similarly, lead-206 has 124 neutrons, lead-207 has 125 neutrons and lead-208 has 126 neutrons.How many neutrons does magnesium have?
12 neutrons
How many total electrons does xenon have?
54 electrons
How many neutrons are in aluminum?
14 neutrons
Is Xenon a neutral atom?
Xenon atoms have 54 electrons and the shell structure is 2.8. The ground state electronic configuration of neutral xenon is [Kr].Can you tell from the periodic table exactly how many neutrons are in an atom?
Because we can find the number of protons and the atomic mass of an atom by looking at its element information in the periodic table, we can calculate the number of neutrons in that atom by subtracting the number of protons from the atomic mass.What is the charge of a proton?
Proton Charge While a proton has a charge of +1, or 1e, an electron has a charge of -1, or -e, and a neutron has no charge, or 0e. 1 elementary charge is equal to: 1.602 x 10^-19 coulombs.