What is the separation of crystallization?
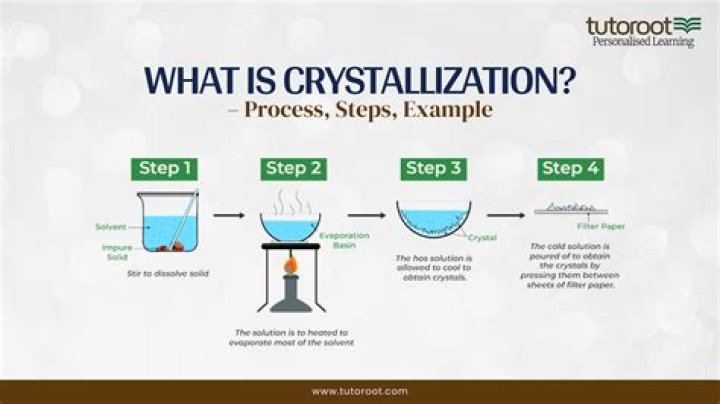
What is the separation of crystallization?
Crystallization is a separation process that makes use of differences in solubility of the components present in the melt or the solution. Crystallization can occur when the solubility of one or more components present in the liquid is exceeded.
What is crystallization phase?
Crystallization is in essence the separation of an ordered crystalline phase from a metastable solution. This means that in a system of a given chemical composition, a stable, protein-rich phase in the form of a crystal, in equilibrium with its growth solution, must exist.
What are the steps during crystallization?
There are five major steps in the recrystallization process: dissolving the solute in the solvent, performing a gravity filtration, if necessary, obtaining crystals of the solute, collecting the solute crystals by vacuum filtration, and, finally, drying the resulting crystals.
Is crystallization a phase change?
Crystallization is a complex phase transition process influenced by a number of parameters, such as temperature, pressure, and concentration, which can serve as an effective approach to materials fabrication.
What happens to particles during crystallisation?
What Happens During a Crystallization. To crystallize an impure, solid compound, add just enough hot solvent to it to completely dissolve it. As the solution cools, the solvent can no longer hold all of the solute molecules, and they begin to leave the solution and form solid crystals.
How are crystals formed in crystallization?
Crystallization or crystallisation is the process by which a solid forms, where the atoms or molecules are highly organized into a structure known as a crystal. Some of the ways by which crystals form are precipitating from a solution, freezing, or more rarely deposition directly from a gas.
What is the principle of crystallization?
The principle of crystallization is based on the limited solubility of a compound in a solvent at a certain temperature, pressure, etc. A change of these conditions to a state where the solu- bility is lower will lead to the formation of a crystalline solid.
What type of substances can be separated by crystallization?
Answer: Crystallization is a method for separating solids from a liquid solution.
What type of change is crystallisation?
Crystallisation is an example of chemical change.
What causes crystallization?
Crystallization is a natural process which occurs as materials solidify from a liquid, or as they precipitate out of a liquid or gas. This can be caused by a physical change, such as a temperature change, or a chemical change such as acidity.
How do crystals form during crystallisation?
Why do crystals form during crystallisation?