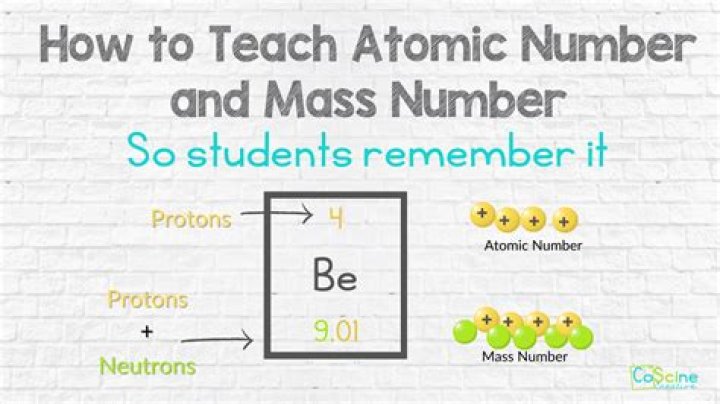
Protons and neutrons have approximately the same mass, about 1.67 × 10−24 grams, which scientists define as one atomic mass unit (amu) or one Dalton. Each electron has a negative charge (−1) equal to the positive charge of a proton (+1). Neutrons are uncharged particles found within the nucleus..
Also, what has the same mass as a proton?
Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron). The positive charge on a proton is equal in magnitude to the negative charge on an electron.
Also, which of the following have approximately the same mass? The nuclear particles, protons and neutrons, are conceived to have a positive electronic charge, and a neutral charge respectively. The protonic mass is 1.672621898(21)×10−27⋅kg . And the neutron mass is 1.674927471(21)×10−27⋅kg , so to a first approx. these masses are equal.
Simply so, which subatomic particles have approximately the same mass?
Answer and Explanation: The two subatomic particles that have the same mass are protons and neutrons. Neutrons and protons each have an atomic mass of about one atomic mass
What is the approximate ratio of the mass of an electron to that of a proton?
The mass of an electron is only about 1/2000 the mass of a proton or neutron, so electrons contribute virtually nothing to the total mass of an atom. Electrons have an electric charge of −1, which is equal but opposite to the charge of a proton, which is +1.
Related Question Answers
What has a mass of 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.Do Protons have mass?
Protons and neutrons have approximately the same mass, about 1.67 × 10-24 grams. Scientists define this amount of mass as one atomic mass unit (amu) or one Dalton. Although similar in mass, protons are positively charged, while neutrons have no charge.What is atomic mass number?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is different for each different isotope of a chemical element.Has there really little mass?
The neutron helps keep the nucleus together. The electron has a negative charge and very little mass (1800 times less than protons and neutrons). Table 1. Relative masses and charges of the particles in an atom.Which particle has the least mass?
Answer - The electron mass has the smallest mass between the basic particles inside the atoms.
- Inside the atom consists of a nucleus containing 3 basic particles namely protons and neutrons, while electrons move around the nucleus.
- The charge of 1 proton is equal to a charge of 1 electron, but the sign is different.
Does an electron have mass?
, whose electric charge is negative one elementary charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton.How is a compound formed?
A compound is a substance formed when two or more elements are chemically joined. Water, salt, and sugar are examples of compounds. When the elements are joined, the atoms lose their individual properties and have different properties from the elements they are composed of.Who discovered Proton?
Rutherford
What is the smallest subatomic particle?
Quarks represent the smallest known subatomic particles. These building blocks of matter are considered the new elementary particles, replacing protons, neutrons and electrons as the fundamental particles of the universe.How are elements identified?
The periodic table can be used to identify an element by looking for the element's atomic number. The atomic number of an element is the number of protons found within the atoms of that element. Therefore, the number of protons can be found by subtracting the number of neutrons from the mass number of the element.Which particles contribute to the mass number and which do not why?
Which particles contribute to the mass number and which do not? Why? Electrons do not affect the mass number but neutrons and protons do. Electrons do not have a mass.Which two particles in an atom are equal in mass?
The number of protons in the nucleus of an atom is known as the atomic number, Z. It is the same as the number of electrons around the nucleus of the electrically-neutral atom. The mass number of an atom is equal to the total number of protons and neutrons.What is a proton mass?
Proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.What percentage of the atom is empty space?
99.9999999 percent
What is the charge and mass of an electron?
Magnitutide of charge: Charge of electron is 1.6022 x 10-19 Coulomb. Mass of electron: Mass of electron is 0.000548597 a.m.u. or 9.1 x 10-31 kg. Symbol of electron: Electron is represented by "e". Location in the atom: Electrons revolve around the nucleus of atom in different circular orbits.What two particles equal a number?
Answer and Explanation: The two particles in the atom that are equal in number are the protons and electrons. The protons are located in the nucleus along with neutrons.What is found outside the nucleus?
Unlike protons and neutrons, which are located inside the nucleus at the center of the atom, electrons are found outside the nucleus. It shows the electron as a particle orbiting the nucleus, similar to the way that planets orbit the sun.Where is proton located?
nucleus
What is the charge of a proton?
Proton Charge While a proton has a charge of +1, or 1e, an electron has a charge of -1, or -e, and a neutron has no charge, or 0e. 1 elementary charge is equal to: 1.602 x 10^-19 coulombs.