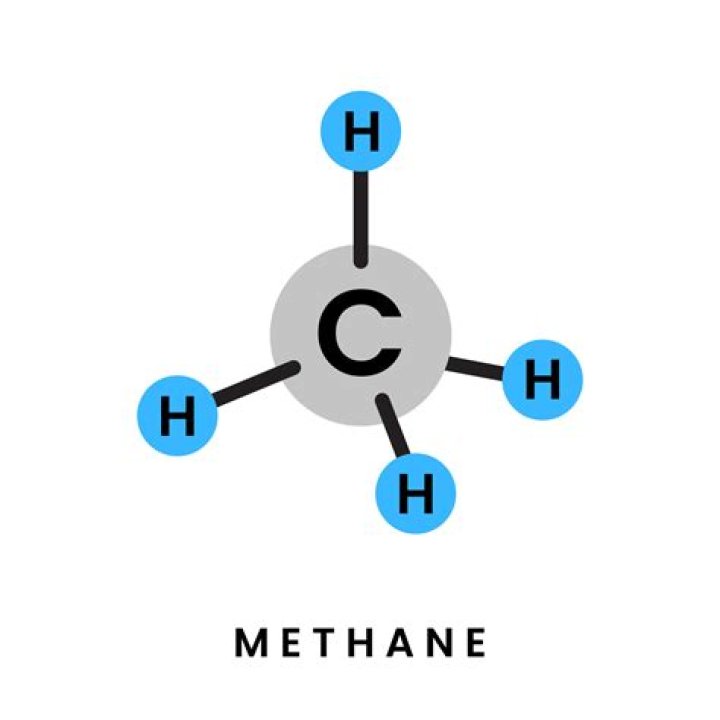
The carbon atom central to the methane molecule has 4 valence electrons and thus needs 4 more electrons from four hydrogen atoms to complete its octet. The hydrogen atoms have a 109 degree bond angle giving the molecule a tetrahedral geometry ..
Similarly one may ask, what type of structure does methane have?
Methane is a compound containing two elements, carbon and hydrogen. It exists naturally as a molecule. Each methane molecule has a central carbon atom joined to and surrounded by four hydrogen atoms.
Also, what is the shape of methane? The shape of methane That is a tetrahedral arrangement, with an angle of 109.5°. Nothing changes in terms of the shape when the hydrogen atoms combine with the carbon, and so the methane molecule is also tetrahedral with 109.5° bond angles.
One may also ask, what contains methane?
Methane, CH4, is a colorless, odorless gas with a wide distribution in nature. It is the principal component of natural gas, a mixture containing about 75% CH4, 15% ethane (C2H6), and 5% other hydrocarbons, such as propane (C3H8) and butane (C4H10). The “firedamp” of coal mines is chiefly methane.
Do humans produce methane?
As for methane (CH4), most humans can't produce it at all. Methane in the body results from microbes called methanogens, which are not bacteria but members of the Archaea kingdom, the oldest life forms on the planet. Only about one-third of humans have methanogens among their gut flora.
Related Question Answers
What is the main source of methane?
Methane Emissions: Natural Sources Methane is also released into the atmosphere by natural processes. Wetlands, termites and the oceans are all natural sources of methane emissions. The methane produced by natural sources is completely offset by natural methane sinks.How methane is created?
Methane is produced at shallow levels (low pressure) by anaerobic decay of organic matter and reworked methane from deep under the Earth's surface. In general, the sediments that generate natural gas are buried deeper and at higher temperatures than those that contain oil.Does every methane molecule look different?
Chemical and Physical Properties of Methane. The carbon atom central to the methane molecule has 4 valence electrons and thus needs 4 more electrons from four hydrogen atoms to complete its octet. The hydrogen atoms have a 109 degree bond angle giving the molecule a tetrahedral geometry .What is h2 called?
H2 is also called molecular hydrogen.It consists of two protons and two electrons. Consequently it is the most common form of Hydrogen because it is stable with a neutral charge. H2 is not a free radical. It is the antioxidant in 'hydrogen-rich' water. H2 is the smallest molecule in the universe.Why is methane bad for the environment?
If methane leaks into the air before being used – from a leaky pipe, for instance – it absorbs the sun's heat, warming the atmosphere. For this reason, it's considered a greenhouse gas, like carbon dioxide.Is methane heavier than air?
Methane [CH4] Natural gas or Methane is also lighter than air, not because it contains helium, which most of it does not, but because it is just less dense than air. It is loaded with Ethane and other gases in addition to methane and thus turns out to be heavier than, or about the same density as air.Why is methane nonpolar?
Why is methane a nonpolar molecule even though it has four polar bonds? ~ Though it does have polar bonds, methane is a nonpolar molecule because its regular tetrahedron shape leads to a symmetrical distribution of the molecules partial charges.What food produces the most methane?
It takes up to 10 pounds of farmed plant foods fed to cows to produce just 1 pound of meat. All that food results in cow-produced methane, a greenhouse gas that's 25 times more potent than carbon dioxide.What is the largest source of methane gas?
Natural gas and petroleum systems are the largest source of CH4 emissions in the United States. Methane is the primary component of natural gas.Can methane kill you?
In severe cases, there may be changes in breathing and heart rate, balance problems, numbness, and unconsciousness. If exposure is large or continues for a longer period it can kill. Skin or eye contact with liquefied methane released under pressure may cause frostbite. Can Methane cause cancer?What is methane made up of?
Methane is a molecule composed of one carbon atom linked to four hydrogen atoms.Is methane a fossil fuel?
Fossil fuels range from volatile materials with low carbon-to-hydrogen ratios (like methane), to liquids (like petroleum), to nonvolatile materials composed of almost pure carbon, like anthracite coal. Methane can be found in hydrocarbon fields either alone, associated with oil, or in the form of methane clathrates.What is the main sources of methane?
There are six major sources of atmospheric methane: emission from anaerobic decomposition in (1) natural wetlands; (2) paddy rice fields; (3) emission from livestock production systems (including intrinsic fermentation and animal waste); (4) biomass burning (including forest fires, charcoal combustion, and firewoodIs methane a clean burning fuel?
Methane is a significant and plentiful fuel which is the principal component of natural gas. Burning one molecule of methane in the presence of oxygen releases one molecule of CO2 (carbon dioxide) and two molecules of H2O (water). Methane's relative abundance and clean burning process makes it a very attractive fuel.What gas is a fart?
Other gases are produced in your digestive tract as the food you eat is broken down. Farts are primarily made of odorless vapors like carbon dioxide, oxygen, nitrogen, hydrogen, and sometimes methane.What state is methane in the Earth's atmosphere?
Atmospheric methane is the methane present in Earth's atmosphere. Atmospheric methane concentrations are of interest because it is one of the most potent greenhouse gases in Earth's atmosphere.What is the shape of ammonia molecule?
trigonal pyramidal
What is the angle of ch4?
The CH4 bond angle is 109.5°. This is the undistorted tetrahedral angle.What is the shape of water molecule?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.