Which base is used in aldol condensation in green synthesis?
Which base is used in aldol condensation in green synthesis?
The Aldol condensation is carried out by mixing freshly ground mixture of the lithium enolate of methyl-3,3-dimethylbutanoate with powdered Page 4 Green Synthesis of Organic compounds aromatic aldehyde in vacuum for 3 days at room temperature.
What is base catalyzed aldol condensation?
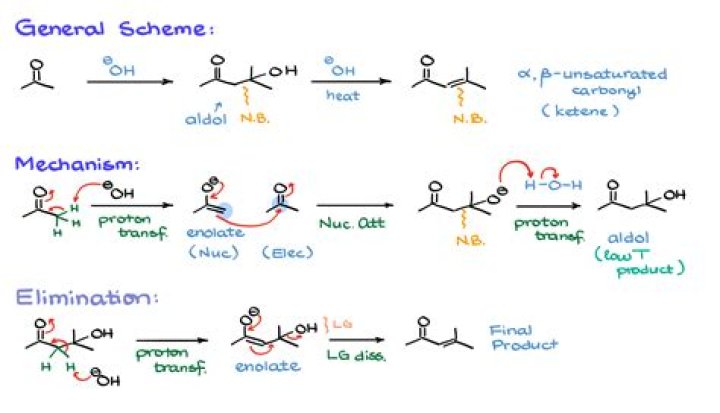
Base catalysed aldol-condensation occurs with propionaldehyde. Aldehydes and ketones having α –H atom are treated with dilute alkali ,two molecules of such compound condense to form β-hydroxy aldehyde and β–hydroxy ketone. The reaction is called as aldol condensation.
What is aldol condensation explain with suitable example?
An aldol condensation is a condensation reaction in organic chemistry in which an enol or an enolate ion reacts with a carbonyl compound to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), followed by dehydration to give a conjugated enone.
Which type of compounds undergo aldol condensation explain with an example?
Aldehydes and ketones having at least one α-hydrogen undergo aldol condensation. The compounds (ii) 2-methylpentanal, (v) cyclohexanone, (vi) 1-phenylpropanone, and (vii) phenylacetaldehyde contain one or more α-hydrogen atoms. Therefore, these undergo aldol condensation.
What does green mean in green aldol?
The word “green” has become synonymous with sustainability. The chemical manufacturing industry uses the term “green chemistry” to mean the design of processes that reduce or eliminate negative environmental impacts, such as the production of hazardous waste.
Which catalyst is used for the green synthesis of benzoin?
Thiamine is a catalyst; it is regenerated each time a benzoin molecule is synthesized. 2) Show how cyanide can replace thiamine in this reaction.
What are base catalyzed reactions?
Base catalyzed: A process that is catalyzed by a base. In the absence of strong acid or strong base, acetone and benzaldehyde do not react. In the presence of a strong base such as NaOH, acetone and benzaldehyde participate in an aldol condensation reaction.
Why is NaOH used in aldol condensation?
NaOH it undergoes self condensation as it contains alpha-hydrogen atom in its compound forming β-hydroxyaldehyde (an aldol) namely 3-Hydroxy butanal. This compound upon further heating will eliminate a molecule of water forming aldol condensation product namely Crotonaldehyde Or But-2-en-al.
What is aldol condensation with Example Class 12?
β-hydroxy aldehyde is commonly known as aldol, and the β-hydroxy ketone is commonly known as ketol. Thus, aldol condensation reaction occurs when aldehydes or ketones have at least one α-hydrogen reaction in the presence of a dilute alkali, e.g., NaOH, to produce β-hydroxy aldehyde or β-hydroxy ketone.
What is aldol condensation explain with example class 12?
Aldehydes containing at least one α–hydrogen atom undergo a reaction in presence of dilute alkali (dilute NaOH, KOH, or Na2CO3) as the catalyst to form β-hydroxy aldehydes (aldol). This gives rise to α,β-unsaturated aldehyde. The overall reaction is called aldol condensation.
Does propanone undergo aldol condensation?
Propanone aldol condensation Propanone is a ketone. Propanone contains three carbon atoms. After aldol condensation of propanone , there are six carbon atoms in the carbon chain. First gives the aldol product with dilute NaOH or KOH.
Which of the following carbonyl compounds undergo aldol condensation?
All carbonyl compounds containing α−H or α−D undergo aldol condensation. In given example, benzaldehyde does not contain α−H to -CHO, hence does not undergo aldol condensation.