Which is a characteristic of a strong acid?
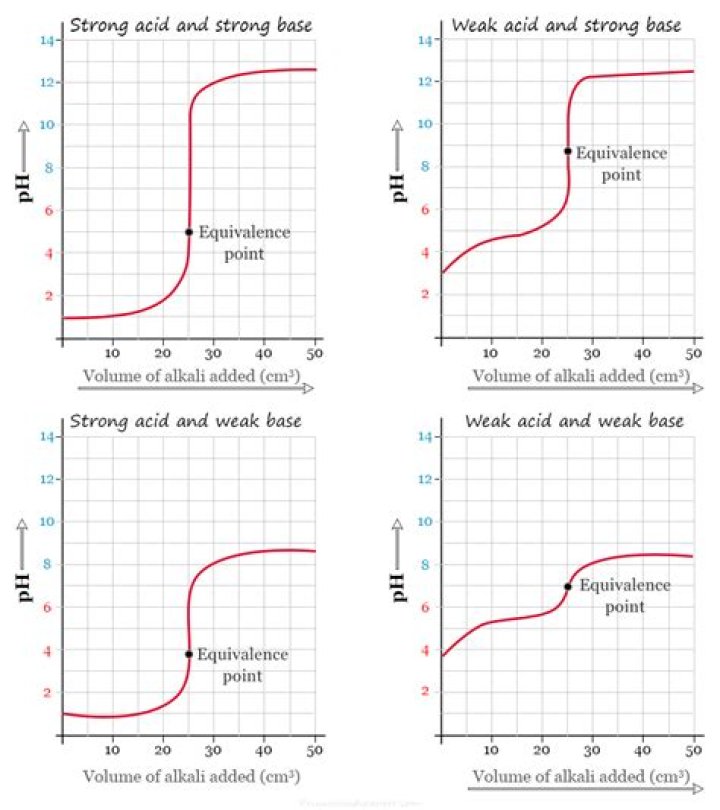
.
Simply so, what is a characteristic of an acid?
General Characteristics of Acids: Sour taste (though you should never use this characteristic to identify an acid in the lab) Reacts with a metal to form hydrogen gas. Increases the H+ concentration in water. Donates H+ ions. Turns blue litmus indicator red.
Also Know, how do you tell if something is a strong or weak acid? If an acid is not listed here, it is a weak acid. It may be 1% ionized or 99% ionized, but it is still classified as a weak acid. Any acid that dissociates 100% into ions is called a strong acid. If it does not dissociate 100%, it is a weak acid.
Keeping this in consideration, what defines a strong acid?
It is very important to know the number of hydrogen ions or protons that can be given off by a particular acid. This determines an acid's strength. A strong acid is any acid that ionizes completely in solutions. This means it gives off the greatest number of hydrogen ions or protons when placed in a solution.
What makes acid strong or weak?
Strong acids completely dissociate into their ions in water, while weak acids only partially dissociate. There are only a few (7) strong acids, so many people choose to memorize them. All the other acids are weak. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).
Related Question AnswersWhat are the 5 properties of acids?
Terms in this set (5)- acids have a sour taste.
- acid formulas start with an "H" contain hydrogen, and some react with active metals in single replacement reaction to give off hydrogen gas.
- acids change the color of certain dyes (acid-base indicators)
What are 3 characteristics of bases?
Bases have these characteristics:- Bitter taste (opposed to sour taste of acids)
- Slimy, or soapy feel on fingers (Slippery)
- Many bases react with acids and precipitate salts.
- Strong bases may react violently with acids.
- Bases turn red litmus paper blue.
- Bases are substances that contain metal oxides or hydroxides.
What is the base?
In chemistry, a base is a chemical species that donates electrons, accepts protons, or releases hydroxide (OH-) ions in aqueous solution. Types of bases include Arrhenius base, Bronsted-Lowry base, and Lewis base.What characteristics do acids and bases have in common?
Acids generally taste sour due to the sour H+ ion; bases taste bitter due to the OH- ion; but they may have other tastes depending on the other part of the molecule. Bases are usually soapy in nature. Acids corrode active metals ; Bases denature protein. Acids have a pH less than 7 ; Bases have a pH greater than 7.What are the properties of strong acids?
Strong and Weak Acids/Bases[edit] A strong acid is an acid which dissociates completely in water. That is, all the acid molecules break up into ions and solvate (attach) to water molecules. Therefore, the concentration of hydronium ions in a strong acid solution is equal to the concentration of the acid.What do bases do to skin?
Strong Bases Bases with a pH greater than 10 can cause chemical burns. Strong bases include, calcium hydroxide, sodium hydroxide and potassium hydroxide. Bases also react with oils on skin and fatty tissue, which can lead to extensive damage to skin and subcutaneous tissue.What are some examples of acids?
Examples of Acids- Hydrochloric acid (HCl) in gastric juice.
- Sulphuric acid (H2SO4)
- Nitric acid (HNO3)
- Carbonic acid in soft drink (H2CO3)
- Uric acid in urine.
- Ascorbic acid (Vitamin C) in fruit.
- Citric acid in oranges and lemons.
- Acetic acid in vinegar.
What is a basic solution?
A basic solution is an aqueous solution containing more OH-ions than H+ions. Examples of common basic solutions include soap or detergent dissolved in water or solutions of sodium hydroxide, potassium hydroxide, or sodium carbonate.What are the 7 strong acids?
There are 7 strong acids: chloric acid, hydrobromic acid, hydrochloric acid, hydroiodic acid, nitric acid, perchloric acid, and sulfuric acid. Being part of the list of strong acids doesn't give any indication of how dangerous or damaging an acid is though.What are weak acids?
A weak acid is an acid chemical which does not dissociate (split into ions) completely in water solution. This means it does not give all its hydrogen ions into the water. Weak acids typically have a pH between 3 and 6. Acetic acid (CH3COOH) and oxalic acid (H2C2O4) are examples of weak acids.What is an example of a weak acid?
A weak acid is an acid that doesn't produce many hydrogen ions when in aqueous solution. Weak acids have relatively low pH values and are used to neutralize strong bases. Examples of weak acids include: acetic acid (vinegar), lactic acid, citric acid, and phosphoric acid.What are the strong bases?
Strong bases are able to completely dissociate in water- LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.