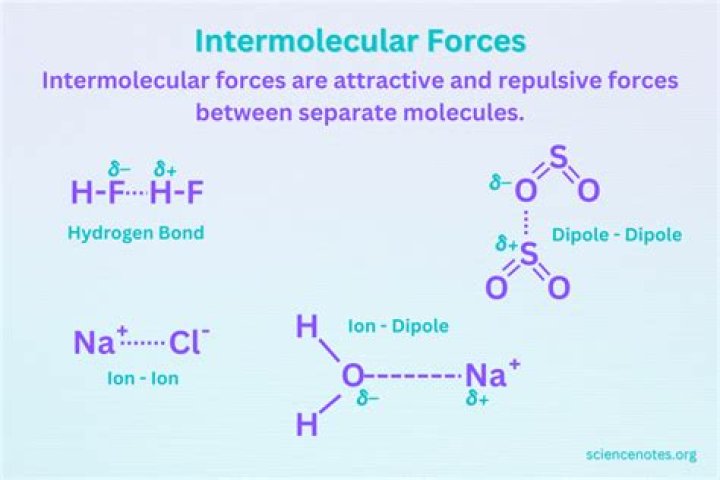
Explanation: Ion-dipole forces are the strongest of the intermolecular forces. Hydrogen bonding is a specific term for a particularly strong dipole-dipole interaction between a hydrogen atom and a very electronegative atom (oxygen, fluorine, or nitrogen)..
Also, which of the following has the strongest intermolecular forces?
From strongest to weakest, the intermolecular forces rank in the following way:
- Strongest: Hydrogen bonding. This occurs when compounds contain #"O"-"H"# , #"N"-"H"# , or #"F"-"H"# bonds.
- Less strong: Dipole-dipole forces.
- Weakest: London Dispersion Forces.
Beside above, which has the strongest dipole? The larger the difference in electronegativities of bonded atoms, the larger the dipole moment. For example, NaCl has the highest dipole moment because it has an ionic bond (i.e. highest charge separation).
Also, what is the strongest intermolecular force present for ch3ch2ch2ch3?
Therefore, CH3CH2CH2CH3 is a nonpolar molecular compound and the only intermolecular. forces are induced dipole forces. Hydrogen bonding is found in situations represented by D-H---A where both the donor atom, D, and the acceptor atom, A, are one of the highly electronegative elements O, N, or F.
Is HCl dipole dipole?
HCl molecules, for example, have a dipole moment because the hydrogen atom has a slight positive charge and the chlorine atom has a slight negative charge. Because of the force of attraction between oppositely charged particles, there is a small dipole-dipole force of attraction between adjacent HCl molecules.
Related Question Answers
What are the types of intermolecular forces?
The three major types of intermolecular interactions are dipole–dipole interactions, London dispersion forces (these two are often referred to collectively as van der Waals forces), and hydrogen bonds.What are intramolecular forces?
An intramolecular force is any force that binds together the atoms making up a molecule or compound, not to be confused with intermolecular forces, which are the forces present between molecules. Chemical bonds are considered to be intramolecular forces, for example.What is intermolecular force?
From Wikipedia, the free encyclopedia. Intermolecular forces (IMF) are the forces which mediate interaction between molecules, including forces of attraction or repulsion which act between molecules and other types of neighboring particles, e.g. atoms or ions.What is van der Waals forces in chemistry?
Van der Waals forces' is a general term used to define the attraction of intermolecular forces between molecules. There are two kinds of Van der Waals forces: weak London Dispersion Forces and stronger dipole-dipole forces.What is a dipole dipole force?
Dipole-dipole forces are attractive forces between the positive end of one polar molecule and the negative end of another polar molecule. Dipole-dipole forces have strengths that range from 5 kJ to 20 kJ per mole. The partially positive end of a polar molecule is attracted to the partially negative end of another.Which substance has the weakest intermolecular forces?
The vapor pressure curves on Table H indicate that propanone has the weakest intermolecular attraction and ethanoic acid has the greatest intermolecular attraction.Is HCL polar or nonpolar?
HCL is neither polar or non-polar. HCL is written wrong, and probably a typographical error. HCl, also known as Hydrogen Chloride is a gas at STP, and is a polar molecule. In HCl, the hydrogen atom is partially positively charged, while the chlorine atom is partially negatively charged.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Does water have dipole dipole forces?
Water has polar O-H bonds. The negative O atoms attract the positive H atoms in nearby molecules, leading to the unusually strong type of dipole-dipole force called a hydrogen bond. Since water has hydrogen bonds, it also has dipole-induced dipole and London dispersion forces.What determines the physical properties of a compound?
The atoms of different elements are held together in compounds by chemical bonds. Bonds help determine the properties of a compound. The properties of a compound depend not only on which atoms the compound contains, but also on how the atoms are arranged.Is co2 dipole dipole?
A molecule like CO2 may be composed of two dipoles, but it has no dipole moment. When molecules have an even charge distribution and no dipole moment, then they are nonpolar molecules. CO2 is a linear molecule, so our dipoles are symmetrical; the dipoles are equal in magnitude but point in opposite directions.Are hydrogen bonds stronger than dipole dipole?
A hydrogen bond is about 10% of the typical covalent bond strength, making it a much stronger attractive force than dipole-dipole and london dispersion.Is ch3oh dipole dipole?
Methanol is a polar molecule (1.69 D), and so it exhibits all three of the van der Waals forces: Keesom forces (dipole-dipole attraction), Debye forces (induced attraction) and London dispersion forces (which all molecules exhibit).Is HBr dipole dipole?
HBr is a polar molecule: dipole-dipole forces. There are also dispersion forces between HBr molecules.What types of intermolecular forces exist in CCl4?
Its strongest intermolecular forces are London dispersion forces. CCl4 is a tetrahedral molecule with a Cl-C-Cl bond angle of 109.5°. The two C-Cl bond dipoles in the plane of the paper have a resultant pointing to the right at an angle of 54.75° from the vertical.Is ch3ch2ch3 polar or nonpolar?
CH3CH2CH3 and CH3CH2OH are somewhat similar in size and complexity, but CH3CH2CH3 is nonpolar while CH3CH2OH is polar with hydrogen bonding. Therefore CH3CH2OH has the strongest forces.What intermolecular forces are present in 1 butanol?
The major intermolecular forces are hydrogen bonding, dipole-dipole interaction, and London/van der Waals forces. All three are found among butanolWhat kind of intermolecular forces exist in ch3ch2ch2oh?
In liquid propanol, CH3CH2CH2OH, which intermolecular forces are present? Dispersion, hydrogen bonding and dipole-dipole forces are present.