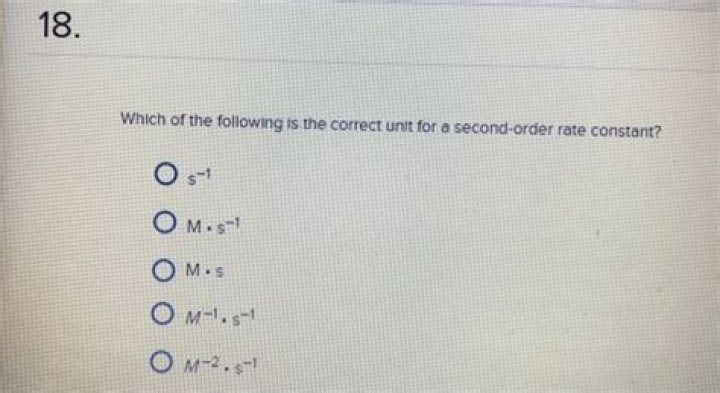
For the units of the reaction rate to be moles per liter per second (M/s), the units of a second-order rate constant must be the inverse (M−1·s−1). Because the units of molarity are expressed as mol/L, the unit of the rate constant can also be written as L(mol·s)..
Also asked, what are the units of the rate constant for a second order reaction?
Two of the same reactant (A) combine in a single elementary step. where k is a second order rate constant with units of M -1 min -1 or M -1 s -1. Therefore, doubling the concentration of reactant A will quadruple the rate of the reaction.
Subsequently, question is, what are the units for rate constant third order reaction? If the reaction is third order, the rate constant should have units of L2 mol-2 s-1.
Also to know is, what are the units for the rate constants of first order and second order reactions?
In first order reactions, the reaction rate is directly proportional to the reactant concentration and the units of first order rate constants are 1/sec. In bimolecular reactions with two reactants, the second order rate constants have units of 1/M*sec.
What is a zero order reaction?
Definition of zero-order reaction : a chemical reaction in which the rate of reaction is constant and independent of the concentration of the reacting substances — compare order of a reaction.
Related Question Answers
What is a second order rate constant?
The simplest kind of second-order reaction is one whose rate is proportional to the square of the concentration of one reactant. A second kind of second-order reaction has a reaction rate that is proportional to the product of the concentrations of two reactants. Such reactions generally have the form A + B → products.What is first order reaction?
A first-order reaction is a reaction that proceeds at a rate that depends linearly on only one reactant concentration.What is rate constant k?
The rate constant, k, is a proportionality constant that indicates the relationship between the molar concentration of reactants and the rate of a chemical reaction. The units of the rate constant depend on the order of reaction.What is the constant k?
The symbol k is a proportionality constant known as the Coulomb's law constant. The value of this constant is dependent upon the medium that the charged objects are immersed in. In the case of air, the value is approximately 9.0 x 109 N • m2 / C2.What is the overall order of the reaction?
The overall order of reaction is the sum of the individual orders of reaction of the reactants and it measures the sensitivity of the reaction to changes in the concentrations of all the reactants. The individual orders of reaction and therefore the overall order of reaction are determined experimentally.What is second order of reaction?
A second order reaction is a type of chemical reaction that depends on the concentrations of one-second order reactant or two first-order reactants. This reaction proceeds at a rate proportional to the square of the concentration of one reactant, or the product of the concentrations of two reactants.What is Molecularity of reaction?
Molecularity. The molecularity of a reaction is defined as the number of molecules or ions that participate in the rate determining step. A mechanism in which two reacting species combine in the transition state of the rate-determining step is called bimolecular.What are the units for zero order reaction?
The rate law for a zero-order reaction is rate = k, where k is the rate constant. In the case of a zero-order reaction, the rate constant k will have units of concentration/time, such as M/s.Why is a first order reaction never completed?
Hence we can say that first order reaction will never complete or will be completed at infinite time and hence the decay graph line will never meet x-axis. Rate constant: We derived the first order rate law and from this rate expression first order rate constant can be derived.What is the rate constant for a first order reaction?
A first-order reaction has a rate proportional to the concentration of one reactant. First-order rate constants have units of sec-1. In other words, a first-order reaction has a rate law in which the sum of the exponents is equal to 1.How do you find the rate constant of a second order reaction?
Second Order Reaction. Second order reactions can be defined as chemical reactions wherein the sum of the exponents in the corresponding rate law of the chemical reaction is equal to two. The rate of such a reaction can be written either as r = k[A]2 , or as r = k[A][B].What is the slope of a zero order reaction?
14.4: Zero-Order Reactions. Because rate is independent of reactant concentration, a graph of the concentration of any reactant as a function of time is a straight line with a slope of −k.What are the units for K?
From the pattern of units we can state that for a reaction of kinetic order n, the units of k are: k = 1/tc^(n-1), remembering that c is the amount per litre expressed in mass or molarity and n is the kinetic order.What affects the rate constant?
An increase in temperature increases the rate constant and hence the rate. An increase in concentration increases the rate but not the rate constant. Temperature affects k and k affects R so, temperature affects both while concentration affects only Rate of reactions.How do you graph a first order reaction?
For a first-order reaction, a plot of the natural logarithm of the concentration of a reactant versus time is a straight line with a slope of −k. For a second-order reaction, a plot of the inverse of the concentration of a reactant versus time is a straight line with a slope of k.What is the unit of rate of reaction?
Reaction rates are usually expressed as the concentration of reactant consumed or the concentration of product formed per unit time. The units are thus moles per liter per unit time, written as M/s, M/min, or M/h.What is the half life for a first order reaction?
The half-life of a chemical reaction, regardless of its order, is simply the time needed for half of an initial concentration of a reactant to be consumed by the reaction. Now, a first-order reaction is characterized by the fact that the rate of the reaction depends linearly on the concentration of one reactant.WHAT IS A in the Arrhenius equation?
About Transcript. The Arrhenius equation is k = Ae^(-Ea/RT), where A is the frequency or pre-exponential factor and e^(-Ea/RT) is the fraction of collisions that have enough energy to react (i.e., have energy greater than or equal to the activation energy Ea) at temperature T.What is the difference between rate and rate constant?
Reaction rate or rate of reaction is the measure of how fast or slow a chemical reaction occurs in the conversion of a reactant into a product while a reaction rate constant gives a comparative amount of the reaction rates of reactants and products. The rate of reaction is influenced by the types of reaction.