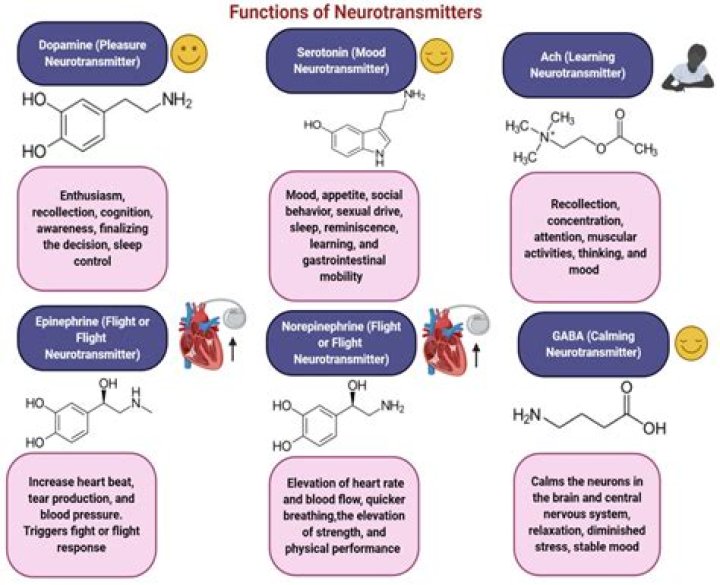
Why the concept of gram atom is useful in chemistry?
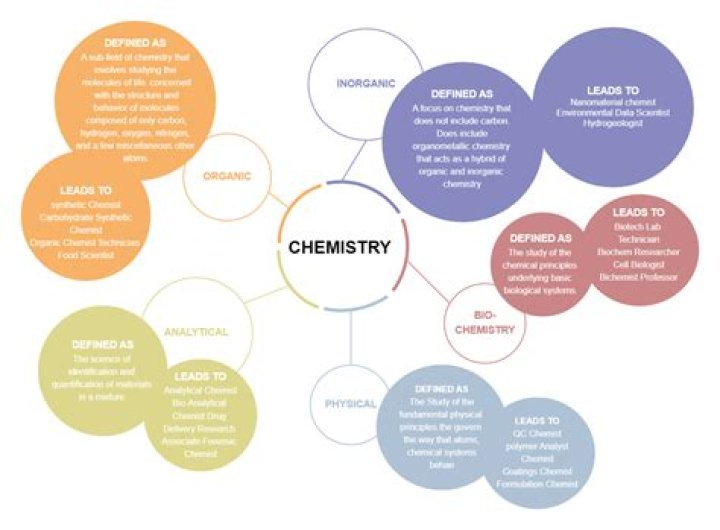
.
Also asked, what is gram atom?
Gram atom is the no. of atoms present in one mole of a substance. of molecules present in one mole of that substance instead of the atoms. And gram ion is the no. of ions present in one mole of that substance.
Similarly, is gram atom the same as mole? Gram atom: The amount or quantity of an element which is numerically equal to its atomic weight (in grams) is termed as gram atom. One mole is defined as the amount of substance of a system which contains as many entities like, atoms, molecules and ions as there are atoms in 12 grams of carbon - 12".
Also question is, what is gram in chemistry?
A gram is a unit of mass in the metric system defined as one thousandth (1 x 10-3) of a kilogram. Originally, the gram was defined as a unit equal to the mass of one cubic centimeter of pure water at 4°C (the temperature at which water has maximum density).
How do you find grams in atoms?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro's number: 6.02 x 10^23.
Related Question AnswersHow many moles are in a gram?
We assume you are converting between moles In and gram. You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles In, or 114.818 grams.What is the meaning of 1 gram molecule?
1 gram molecule means 1 mole of molecules. 1 gram ion means 1 mole of ions. Similarly, 2 gram atom means 2 moles of atoms. So, this is the name of mole.How many grams are in a atom?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro's number: 6.02 x 10^23.What is 1 gram atom of nitrogen?
1 gram- atom of nitrogen corresponds to mass of one mole of the nitrogen atom is grams that is one gram -atom of nitrogen represents 28 gms of nitrogen. It is also equal to 6.023 x 1023 N atoms, but not 6.023 x1023 N2 molecules.What is the weight of 5 gram atoms of nitrogen?
So I think the answer is 56 g. As nitrogen is diatomic it's GMM becomes 28 g.What is in a mole?
The mole (symbol: mol) is the unit of measurement for amount of substance in the International System of Units (SI). A mole of a substance or a mole of particles is defined as exactly 6.02214076×1023 particles, which may be atoms, molecules, ions, or electrons. In short, for particles 1 mol = 6.02214076×1023.What is an example of 1 gram?
Here are common examples of objects that have about one gram of mass: A small paperclip. A thumbtack. A piece of chewing gum.What is a gram example?
A gram is a unit of measure in the metric system that equals one thousandth of a kilogram. An example of a gram is . 035 ounces or four pinches of salt. Gram is defined as a nickname for a grandmother. An example of gram is your mom's mom.Who invented kg?
The word kilogramme was written into French law in 1795, in the Decree of 18 Germinal, which revised the provisional system of units introduced by the French National Convention two years earlier, where the gravet had been defined as weight (poids) of a cubic centimetre of water, equal to 1/1000 of a grave.Where is the gram?
The gram (alternative spelling: gramme; SI unit symbol: g; Latin: gramma, from Greek γράμμα, grámma) is a metric system unit of mass.| Gram | |
|---|---|
| Imperial units U.S. customary | 0.0353 ounces |