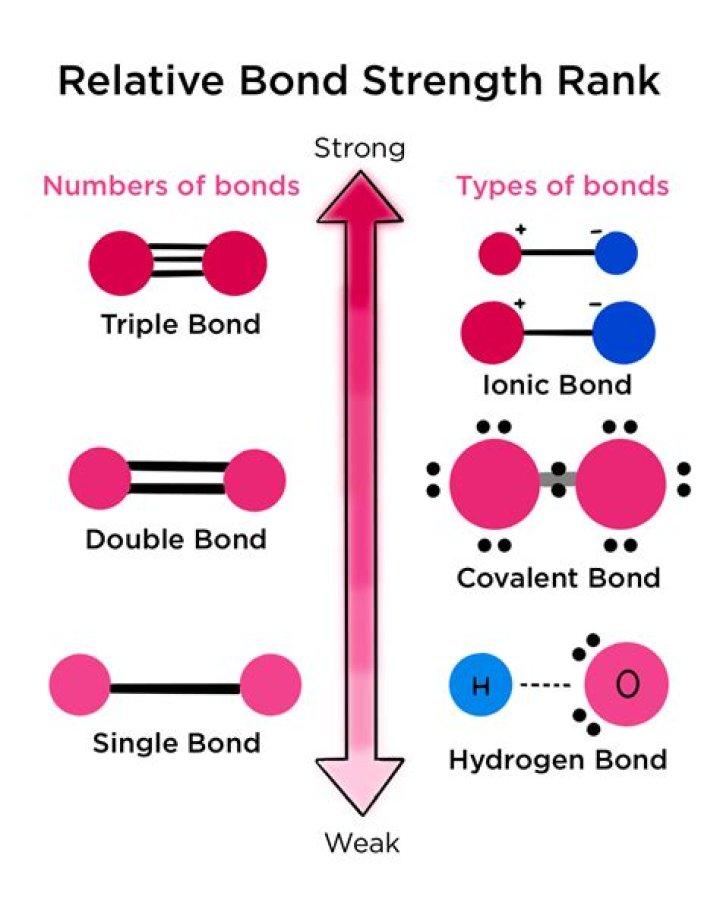
Is triple bond the strongest?
.
In respect to this, why is a triple bond the strongest?
Triple bonds are stronger than double bonds due to the the presence of two [latex]pi[/latex] bonds rather than one. Each carbon has two sp hybrid orbitals, and one of them overlaps with its corresponding one from the other carbon atom to form an sp-sp sigma bond.
Likewise, why is a triple bond stronger than a single bond? The more electrons that are shared between atoms, the stronger the bond. The more electrons that are shared between atoms, the stronger the bond. Single bonds have two electrons shared, double bonds have 4 electrons shared and triple bonds have 6 electrons shared. Thus triple bonds are the strongest.
Secondly, which is the strongest bond?
Answer: Covalent bond is the strongest bond. Answer: There are a variety of ways atoms bond to one another.
Are triple bonds the most stable?
Now,stronger the bond between two atoms, stabler(more stable) the molecule. Hence, triple bond is more stable. In terms of strength, In covalent molecules, more is the sharing of electrons between atoms, stronger is the a single bond 2 electrons are shared,4 in double bond and 6 in triple bond.
Related Question AnswersWhich bond is stronger sigma or pi?
Pi Bonds "π" A pi bond is a weaker chemical covalent bond than a sigma bond (since π bonds have a smaller overlap between the orbitals), but when it is put with a sigma bond it creates a much stronger hold between the atoms, thus double and triple bonds are stronger then single bonds.What is the weakest bond?
A Chemical bond is technically a bond between two atoms that results in the formation of a molecule , unit formula or polyatomic ion. The weakest of the intramolecular bonds or chemical bonds is the ionic bond. The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.How many electrons are in a triple bond?
One covalent bond contains 2 electrons, one from each atom. A triple bond indicates that, well, there are three total covalent bonds in that one bond. So, 3 bonds x 2 electrons per bond would give you 6 total electrons.Does o2 have a triple bond?
Oxygen tends not to form triple bonds due to formal charge reasons. If oxygen starts with 6 electrons and forms a triple bond, then it has 2 lone pair electrons. Using the formal charge formula, 6 - (2+6/2) = 1.What is a triple bond called?
A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. In skeletal formula the triple bond is drawn as three parallel lines (≡) between the two connected atoms.Which bond is stronger CC or CO?
How is the C-O bond stronger than the C-C bond, but the C-N bond is weaker than both of these bonds? It makes sense that C-O is stronger than C-C. The difference in electronegativity is greater which increases ionic character while the oxygen atom is smaller, which increases orbital overlap.Is a covalent bond strong or weak?
Covalent and ionic bonds are both typically considered strong bonds. However, other kinds of more temporary bonds can also form between atoms or molecules. Two types of weak bonds often seen in biology are hydrogen bonds and London dispersion forces.What does bond strength depend on?
In ionic bonds, charge and distance are the two factors that affect the strength of the bond. The more electrons transferred, the stronger the bond.What are some examples of ionic bonds?
Ionic bond examples include:- LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.



