What causes emulsion
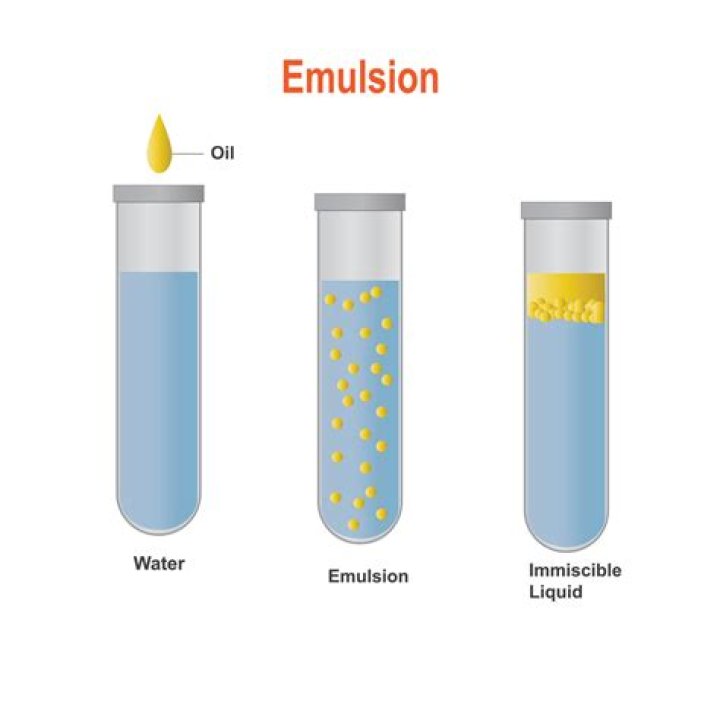
An emulsion is formed when two nonsoluble liquids (e.g., an oil and water) are agitated together to disperse one liquid into the other, in the form of drops. Emulsions can either be oil-in-water (O/W) or water-in-oil (W/O), depending on whether the continuous phase is the water or the oil, respectively.
What causes emulsion separation?
Emulsions contain large areas of fluid interfaces that are thermodynamically unstable. … The main mechanism which leads to phase separation of emulsions is droplet coalescence, where drops merge together to reduce the total interfacial area present.
How do you get emulsion?
Emulsion sauces are made by mixing two substances that don’t normally mix. To do this, you have to break one of them into millions of miniscule droplets and suspend those droplets in the other substance by vigorously whisking, or better yet, blending them in a blender or food processor.
What causes oil emulsion?
Formation of emulsions Crude oil emulsions form when oil and water (brine) come into contact with each other, when there is sufficient mixing, and when an emulsifying agent or emulsifier is present. The amount of mixing and the presence of emulsifier are critical for the formation of an emulsion.What ingredient creates emulsion?
An emulsion is a uniform mixture of two unmixable liquids like oil and water, using agitation from whisking or blending to create a uniform suspension. Depending on how the emulsion is formed, the agitation makes either small oil droplets or water/vinegar droplets.
What causes coalescence in emulsion?
28.6. Coalescence of emulsions is an irreversible process by which two or more droplets merge during contact to form a single daughter droplet. The driving force for emulsion coalescence is the thinning and disruption of the liquid film between the droplets (Fig. 28.9).
What causes flocculation in emulsion?
Flocculation is when the emulsion droplets aggregate and thereby form larger units. Coalescence is when smaller droplets merge together forming a larger droplet. This occurs when droplets come in contact with each other and the interfacial film is ruptured. Eventually the phase separation will happen.
What causes emulsions to crack or cream?
The rise of dispersed particles to the surface of an emulsion is referred to as creaming, which occurs due to density differences between the dispersed particles and the serum phase.How do you fix a broken emulsion?
Fixing any broken egg-based emulsion requires the same method: Create a new emulsion, then whisk the broken one into it. You can do this by placing a teaspoon of lemon juice (or water) in a clean bowl and adding a small amount of the broken emulsion, whisking to form another, stable emulsion.
What emulsified fat?Fat emulsification is the process of increasing the surface area of fats in the small intestine by grouping them into small clusters. This is the responsibility of bile, a liquid created by the liver and stored in the gallbladder. Actual digestion of the fats is then accomplished by lipase, an enzyme from the pancreas.
Article first time published onWhat is emulsion paint?
Emulsion paint is water-based paint, which contains small polymer particles that have pigments inside. The particles containing pigment are suspended in water. After the paint dries, the particles combine, producing a film of paint on the wall.
What do you mean by emulsion?
emulsion, in physical chemistry, mixture of two or more liquids in which one is present as droplets, of microscopic or ultramicroscopic size, distributed throughout the other.
How do you make emulsion paint?
- Pour calcium carbonate into a bucket. …
- Pour 10 litres of water on the calcium carbonate in the bucket.
- Mix the ingredients together .
- Add your desired colour to the mixture.
- Add 1kg of P.V.A to the mixture.
- Stir the mixture very well.
- Add nitrosol powder to water and mix thoroughly.
What ingredient helps to stabilize an emulsion?
To prevent the mixture from separating substances called emulsifiers can be added. These help to form and stabilise the emulsions, preventing or slowing the water and fat/oil from separating.
Is Vinegar an emulsion?
A simple vinaigrette, the combination of oil and vinegar, is an example of a temporary emulsion because the two liquids only briefly stay together before re-separating. On the other had, mayonnaise, the combination of oil, egg yolks, and lemon juice or vinegar is an example of a permanent emulsion.
What are some natural emulsifiers?
- beeswax. Beeswax is extremely beneficial for the body by the way it keeps the skin hydrated. …
- candelilla wax. …
- carnauba wax. …
- rice bran wax. …
- essential oil emulsifiers. …
- how do emulsifiers work with essential oils. …
- Check Out a Few of My Other Favorite Posts.
How can you prevent emulsion from flocculation?
Emulsions also contain emulsifiers – materials that concentrate at the phase interface to lower the interfacial tension. Emulsifiers reduce the energy required to break the dispersed phase into droplets and prevent them from coalescing by generating a repulsive force or a physical barrier between them.
What affects emulsion stability?
The extent of emulsion stability is determined by various factors such as particle size, particle size distribution, density between the dispersed and continuous phases as well as the chemical integrity of the dispersed phase [26].
How do I stop my emulsion from cracking?
emulsion are not stored properly which leads to microbial contamination on to the emulsion, hence the microbes get deteriorate the emulsion and leads to cracking of emulsion. So use preservatives to prevent the microbial growth on to emulsion.
How does pH affect emulsion?
For batch emulsions prepared at pH 3.4, oil-in-water (o/w) emulsions are formed that are stable to coalescence but exhibit creaming. Below pH 3.3, however, these emulsions are very unstable to coalescence and rapid phase separation occurs just after homogenization (pH-dependent).
What is the defect of emulsion?
Insufficient amount of emulsifier. Precipitation of water-soluble emulsifiers. pH disbalance (too high a concentration of base or acid can make emulsifiers disfunctional)
Why do emulsions fail?
Why do emulsions break? Making an emulsion is fairly easy, but it can be a little delicate. Often if the temperature is too high or the olive oil is added too quickly then the mixture can lose its ability to hold together. When this happens, the emulsification has “broken” or “separated.”
Why is my mayonnaise oily?
Cause: There is no longer enough water tu suspend and separate the oil droplets. Solution: Quickly whisk in 5 to 10 ml (1 to 2 teaspoons) of water, vinegar or lemon juice. The mayonnaise will thin out a bit and lose it’s oily appareance. Any remaining oil can be added at this point.
Why is my mayonnaise not emulsifying?
When too much oil is added too fast, it doesn’t emulsify with the egg yolks. Instead of a smooth spread, you actually end up with a sauce that’s broken and curdled-looking. Follow this tip: Add the oil gradually. Start with a teaspoon or two, blend, add another couple teaspoons, then blend again.
What is emulsion cracking?
The Westlake Environmental Emulsion Cracking System (ECS) is highly efficient in removing free, dispersed and emulsified hydrocarbon products from a waste stream. … Lowering the pH of the incoming water destabilizes the petroleum hydrocarbon oil water ionic bond, allowing the oil to become free.
How do you reverse emulsion?
Use gravity to separate your oil and water emulsion. Centrifuge the two substances together. The heavier liquid will reach the bottom first and stay there, while the lighter of the two will remain in a layer on the top.
What is instability emulsion?
There are three types of instability in emulsions: (1) flocculation, (2) creaming, and (3) coalescence. Flocculation occurs when there is an attractive force between the droplets, so they form flocs. … Creaming occurs when the droplets rise to the top of the emulsion under the influence of buoyancy.
Why must fat be emulsified?
Emulsification is important for the digestion of lipids because lipases can only efficiently act on the lipids when they are broken into small aggregates. Lipases break down the lipids into fatty acids and glycerides.
What enzymes break down triglycerides?
lipase, any of a group of fat-splitting enzymes found in the blood, gastric juices, pancreatic secretions, intestinal juices, and adipose tissues. Lipases hydrolyze triglycerides (fats) into their component fatty acid and glycerol molecules.
What is the emulsifier in our body?
An emulsifier is a substance that functions to stabilize an emulsion. … Bile is a substance that works as an emulsifier in the small intestine. Bile is produced by the liver and secreted into the small intestine by the gallbladder, and it functions to emulsify water and fat in the small intestine.
Why is paint called emulsion?
Emulsion is water-based paint, with vinyl or acrylic resins added to make the finish hardwearing. This results in varying degrees of sheen – generally, the shinier the finish, the tougher the paint. Use for interior walls and ceilings. Matt: Gives a matt, non-shiny finish.



