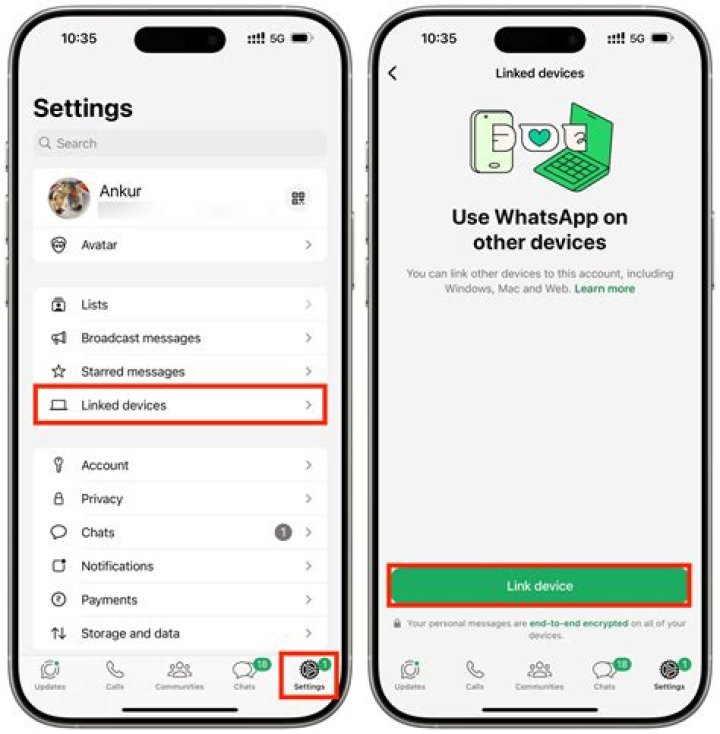
What is a hydrogen particle made of?
What is a hydrogen particle made of?
The hydrogen atom consists of a proton and an electron, and has a spherical symmetry that can most easily be studied using a spherical polar coordinate frame.
How many particles does hydrogen contain?
A hydrogen, nitrogen, or oxygen molecule, consists of two identical atoms of each of those respective elements. Therefore, a hydrogen molecule’s mass is 2 amu, oxygen is 32 amu and nitrogen is 28 amu.
What all contains hydrogen?
Hydrogen combines with other elements to form numerous compounds. Some of the common ones are: water (H2O), ammonia (NH3), methane (CH4), table sugar (C12H22O11), hydrogen peroxide (H2O2) and hydrochloric acid (HCl). Hydrogen has three common isotopes. The simplest isotope, called protium, is just ordinary hydrogen.
Why H+ is known as proton?
Formation of H+ ion: When the hydrogen atom loses its one electron from the valence (outermost) shell results in the formation of hydrogen(H+) ion. Hydrogen ion is also called proton because it having only one proton inside the nucleus of an atom . H+ ion does not contain electrons or neutrons.
How many electrons are found in hydrogen?
one electron
Hydrogen has one one proton and one electron; the most common isotope, protium (1H), has no neutrons.
Is hydrogen gas H2 or H?
Hydrogen has a molar mass of 1 and it’s molecular formula is H2. Hydrogen, H, is the lightest element with the atomic number 1. It is a colorless, odorless, tasteless, and highly flammable gas with the molecular formula H2.
What form is hydrogen found in?
Most hydrogen is produced by heating natural gas with steam to form syngas (a mixture of hydrogen and carbon monoxide). The syngas is separated to give hydrogen….
| Discovery date | 1766 |
|---|---|
| Origin of the name | The name is derived from the Greek ‘hydro’ and ‘genes’ meaning water forming. |
| Allotropes | H2 |
What H2 means?
hydrogen gas
H2, the chemical formula for hydrogen gas (dihydrogen) Deuterium (Hydrogen-2, H-2, 2H), the isotope of hydrogen with one proton, one neutron, and one electron.
Why is H2 formed?
(1) collision of an H-atom with a grain; (2) thermal accommodation and adsorption; (3) retention on the grain surface until another H-atom arrives and is adsorbed; (4) H-atom migration to a reaction site; (5) recombination to form H2; and (6) ejection of this newly formed H2 molecule, possibly in an excited state.
Is hydrogen a proton?
1
Hydrogen/Atomic number
Is hydrogen just a proton?
A hydrogen ion is formed when a hydrogen atom loses an electron and therefore becomes positively charged (it has a charge of +1). A hydrogen atom is therefore often referred to as just a proton, as it is left with only one proton and no electrons, as a H atom only has one of each.