The Boltzmann constant (kB or k), named after its discoverer, Ludwig Boltzmann, is a physical constant that relates the average relative kinetic energy of particles in a gas with the temperature of the gas. The Boltzmann constant has the dimension energy divided by temperature, the same as entropy..
Similarly one may ask, what is K in PV NkT?
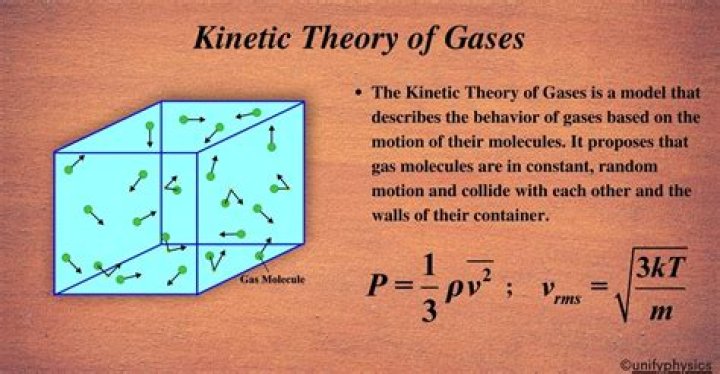
The ideal gas law can be written in terms of the number of molecules of gas: PV = NkT, where P is pressure, V is volume, T is temperature, N is number of molecules, and k is the Boltzmann constant k = 1.38 × 10–23 J/K. The number of molecules in a mole is called Avogadro's number NA, NA = 6.02 × 1023 mol−1.
Also Know, what is K in Boltzmann's formula? (1) where kB is the Boltzmann constant (also written as simply k) and equal to 1.38065 × 10−23 J/K. In short, the Boltzmann formula shows the relationship between entropy and the number of ways the atoms or molecules of a thermodynamic system can be arranged.
Besides, what is K in 3 2kT?
The temperature of a gas can be related to the internal motion of the molecules. The internal energy U of a gas is defined as U = NKE and thus PV = 2=3U. The relation between the mean kinetic energy and the temperature is given by KE = 3=2kT, where. k = R=NA is the Boltzmann constant: k = 1:3807 10-23J=K.
What does kinetic theory explain?
Kinetic theory or kinetic theory of gases attempts to explain overall properties of gases, such as pressure, temperature, or volume, by considering their molecular composition and motion. Instead, pressure is caused by the molecules colliding with each other and their container.
Related Question Answers
What is K value?
Values of k. Units. 1.380649×10−23. J⋅K−1.What is the value of R?
The value of the gas constant 'R' depends on the units used for pressure, volume and temperature. R = 0.0821 liter·atm/mol·K. R = 8.3145 J/mol·K. R = 8.2057 m3·atm/mol·K. R = 62.3637 L·Torr/mol·K or L·mmHg/mol·K.What is the value of k in Boyle's law?
kB=1.38x10^-23J/K , the Boltzmann constant. The value of R is 8.31J/mol. K So, constant in Boyle's law for same gas at same absolute temperature depends on the amount through mole number. You may take nNo=N==number of molecules.What is Z in thermodynamics?
The compressibility factor (Z), also known as the compression factor or the gas deviation factor, is a correction factor which describes the deviation of a real gas from ideal gas behaviour. It is a useful thermodynamic property for modifying the ideal gas law to account for the real gas behaviour.What is K in kinetic energy?
Kinetic Energy Formula. The Kinetic energy is the energy that an object has due to its motion. Ek, is the energy of a mass, m, in motion, v2. Ek = 1/2 mv2.What is the value of k in physics?
The symbol k is a proportionality constant known as the Coulomb's law constant. The value of this constant is dependent upon the medium that the charged objects are immersed in. In the case of air, the value is approximately 9.0 x 109 N • m2 / C2.What is a in van der Waals equation?
The van der Waals equation of state approaches the ideal gas law PV=nRT as the values of these constants approach zero. The constant a provides a correction for the intermolecular forces. Constant b is a correction for finite molecular size and its value is the volume of one mole of the atoms or molecules.What is the R in PV nRT?
The ideal gas law is: pV = nRT, where n is the number of moles, and R is universal gas constant. The value of R depends on the units involved, but is usually stated with S.I. units as: R = 8.314 J/mol·K.What is RMS velocity?
The root mean square velocity (RMS velocity) is a way to find a single velocity value for the particles. The average velocity of gas particles is found using the root mean square velocity formula: μrms = (3RT/M)½What is rms speed?
The root-mean-square speed is the measure of the speed of particles in a gas, defined as the square root of the average velocity-squared of the molecules in a gas. The root-mean-square speed takes into account both molecular weight and temperature, two factors that directly affect the kinetic energy of a material.What is the formula for average kinetic energy?
The average kinetic energy (K) is equal to one half of the mass (m) of each gas molecule times the RMS speed (vrms) squared.What is the constant R?
The value of the gas constant 'R' depends on the units used for pressure, volume and temperature. R = 0.0821 liter·atm/mol·K. R = 8.3145 J/mol·K. R = 8.2057 m3·atm/mol·K. R = 62.3637 L·Torr/mol·K or L·mmHg/mol·K.What is thermal voltage?
The Thermal Voltage is the relationship between the flow of electric current and electrostatic potential across a p-n junction. The thermal voltage depends on absolute temperature T. The formula for Thermal Voltage is: VT=(k•T)/q.Do all gas molecules have the same kinetic energy?
Gases consist of particles (molecules or atoms) that are in constant random motion. The average kinetic energy of gas particles is proportional to the absolute temperature of the gas, and all gases at the same temperature have the same average kinetic energy.What is KBT physics?
The Boltzmann constant (kB or k), named after its discoverer, Ludwig Boltzmann, is a physical constant that relates the average relative kinetic energy of particles in a gas with the temperature of the gas. The Boltzmann constant is defined to be exactly 1.380649×10−23 J/K.Does the average translational kinetic energy of 3 2 kT applies to all ideal gas?
The internal energy of an ideal gas The result above says that the average translational kinetic energy of a molecule in an ideal gas is 3/2 kT. There is also no energy associated with bonds between atoms in molecules, because there are no bonds in a monatomic gas.How do we get thermal energy?
Thermal energy (also called heat energy) is produced when a rise in temperature causes atoms and molecules to move faster and collide with each other. The energy that comes from the temperature of the heated substance is called thermal energy.Why is entropy J K?
It determines that thermal energy always flows spontaneously from regions of higher temperature to regions of lower temperature, in the form of heat. Thermodynamic entropy has the dimension of energy divided by temperature, which has a unit of joules per kelvin (J/K) in the International System of Units.What are the three laws of thermodynamics?
The first law, also known as Law of Conservation of Energy, states that energy cannot be created or destroyed in an isolated system. The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero.