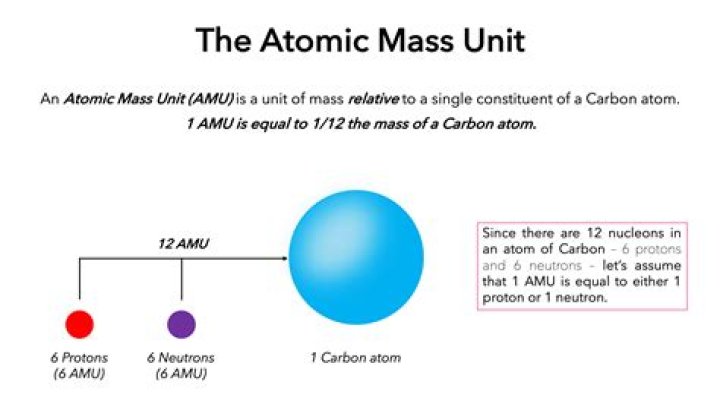
explanation : one atomic mass unit or, amu is equal to one-twelfth of the mass of Carbon - 12 isotope. hence, one atomic mass unit is numerically equivalent to 1g/mol. actually, atomic mass unit or a.m.u is the mass of one nucleon (either a single proton or neutron)..
Simply so, what is the unit for atomic mass?
AMU
Secondly, what is atomic mass unit explain with example? An atomic mass unit is defined as a mass equal to one twelfth the mass of an atom of carbon-12. The mass of any isotope of any element is expressed in relation to the carbon-12 standard. For example, one atom of helium-4 has a mass of 4.0026 amu. An atom of sulfur-32 has a mass of 31.972 amu. Figure 1.
Besides, what do you mean by 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.
Is atomic mass unit a SI unit of an atomic mass?
Units of Atomic Mass; “u,” “Da,” “amu,” and “mmu” The unified atomic mass unit (unit symbol: u) is a non-SI unit of mass, defined as one-twelfth the mass of a single 12C atom in its ground state. Therefore, both the unified atomic mass unit and dalton are authorized units for mass of ions and molecules.
Related Question Answers
How do we find atomic mass?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. Example: Find the atomic mass of an isotope of carbon that has 7 neutrons. You can see from the periodic table that carbon has an atomic number of 6, which is its number of protons.What is the atomic mass of all the elements?
The elemenents of the periodic table sorted by atomic mass
| Atomic Mass | Name chemical element | Symbol |
| 83.8 | Krypton | Kr |
| 85.4678 | Rubidium | Rb |
| 87.62 | Strontium | Sr |
| 88.9059 | Yttrium | Y |
What is atomic mass and mass number?
Atomic mass is the weighted average mass of an atom of an element based on the relative natural abundance of that element's isotopes. The mass number is a count of the total number of protons and neutrons in an atom's nucleus. Is Amu equal to g mol?
The mass of a single atom of an element [amu] is numerically equal to the mass [g] of 1 mol of that element, regardless of the element.Is atomic mass unit a practical unit?
Yes, amu is a practical unit of mass which is used in atomic and nuclear physics. An atomic mass unit or amu is one twelfth of the mass of an unbound atom of carbon-12.What is 1 amu or 1 U?
1 amu or 1 atomic mass unit is equals to the 1/12 of the mass of a carbon atom. U is the standard unit of measure for designating the vertical usable space, or height of racks (metal frame designed to hold hardware devices) and cabinets (enclosures with one or more doors).What is the mass of 1 amu in grams?
One AMU is equivalent to 1.66 x 10-24 grams.How do you find the value of one AMU?
Believe it or not, mathematically, moles and Avogadro's constant bring us back to atomic mass units. Dividing both sides by Avogadro's constant, we find that 1 amu is equal to 1.66 * 10^-27 kg. This means that atomic mass units can be easily converted to kilograms or grams. For example, carbon-12 has a mass of 12 amu.What is difference between AMU and Gram?
The main difference between amu and grams is that amu is used to express the mass in atomic level whereas gram is used as a metric unit of mass.Why is Amu needed?
Why do we need this unit? We need the amu to establish the value of Avogadro's number—the number of particles in one mole. One amu is currently defined as one 12th the mass of one atom of C-12*. By definition the number of amu that equals exactly one gram is Avogadro's number.How much AMU is an electron?
An electron actually weighs about 0.00054 amu. This is so much less than the mass of a proton or a neutron that the mass of the electrons is ignored in determining the mass of an atom.What does U mean in chemistry?
The unified mass unit or dalton (symbol: u, or Da or AMU) is a standard unit of mass that quantifies mass on an atomic or molecular scale (atomic mass). One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.What is a mole of an element?
One mole (abbreviated mol) is equal to 6.022×1023 molecular entities (Avogadro's number), and each element has a different molar mass depending on the weight of 6.022×1023 of its atoms (1 mole). The molar mass of any element can be determined by finding the atomic mass of the element on the periodic table.How is AMU 931 MeV?
If we divide above result by electronic charge we get energy in electron volt. Now if we further divide it by million, the result will be 931 MeV. Thus 931 MeV is equivalent to 1 amu mass.What is atomic mass unit class 9?
Class 9 Chemistry Atoms and Molecules. Atomic mass. Atomic mass. Atomic mass refers to the mass of an atom. It depicts how many times an atom of an element is heavier than one-twelth (1/12th) the mass of one atom of carbon-12 of mass of one carbon atom.Is Dalton same as AMU?
1 Answer. The atomic mass unit (amu) was not renamed to dalton (Da). These are different, albeit related, units. Actually, 'dalton' and 'unified atomic mass unit' are alternative names for the same unit, equal to 1/12 times the mass of a free carbon-12 atom, at rest and in its ground state, i.e.What is atomic mass in Hindi?
?????? ????????? ???? ?? , ??????? , ?????? (what is atomic mass in hindi) ???? ?? ?????? ?? ????? ??? ?? ???? ???? ?? , ?????????? , ??????? , ?????????? ?? ???? ???? ?? ????????? ?? ??? ?? ?????? ????????? ???? ???Why is the atomic mass in a fraction?
The atomic masses of most elements are fractional because they exist as a mixture of isotopes of different masses. Most elements occur as a mixture of isotopes of different masses. The fractional atomic masses arise because of this mixture. mass = total mass of all atoms/number of atoms.