Where are hydrogen bonds used?
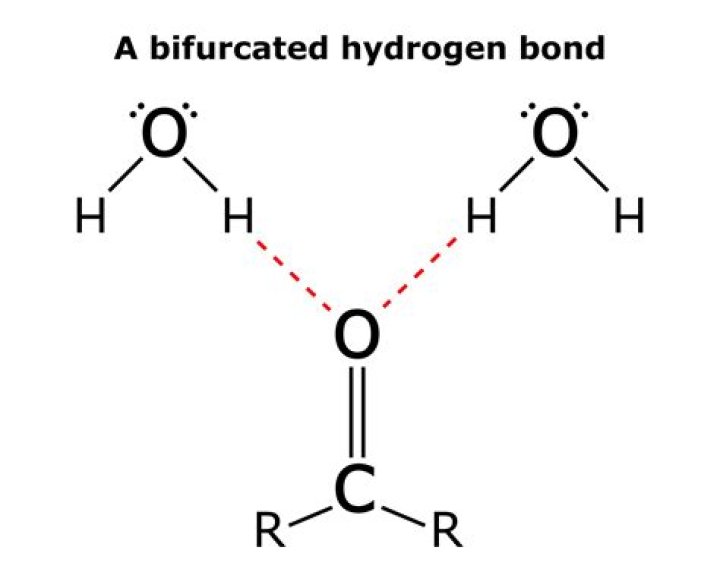
.
Keeping this in consideration, where are hydrogen bonds found?
A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom.
how does a hydrogen bond form? A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. Hydrogen has one proton and one electron. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.
Then, what are some examples of hydrogen bonds?
Examples of Hydrogen Bonds
- water (H2O): Water is an excellent example of hydrogen bonding.
- chloroform (CHCl3): Hydrogen bonding occurs between hydrogen of one molecule and carbon of another molecule.
- ammonia (NH3): Hydrogen bonds form between hydrogen of one molecule and nitrogen of another.
How does hydrogen bonding work?
Hydrogen bonding is the bond (or more accurately, an intermolecular attraction) between a hydrogen atom bound to a highly electronegative atom like Fluorine, Oxygen or Nitrogen. When it bonds with a hydrogen atom, this high charge density causes it to pull the bonded electrons towards itself, creating a dipole.
Related Question AnswersWhat type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.What are the characteristics of hydrogen bonds?
Such properties include its relatively high melting and boiling point temperatures, accounted for by the collection of intermolecular forces between water molecules. Thus the hydrogen bonds require a high input of energy in order to break the bonds between molecules.What are the 3 most important properties of water?
The main properties of water are its polarity, cohesion, adhesion, surface tension, high specific heat, and evaporative cooling. A water molecule is slightly charged on both ends. This is because oxygen is more electronegative than hydrogen.What do you need for hydrogen bonding?
There are two requirements for hydrogen bonding. Two Requirements for Hydrogen Bonding: First molecules has hydrogen attached to a highly electronegative atom (N,O,F). Second molecule has a lone pair of electrons on a small highly electronegative atom (N,O,F).How many hydrogen bonds are in A and T?
Two hydrogen bondsWhy are hydrogen bonds important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.Is h2o a hydrogen bond?
In H2O molecule, two water molecules are bonded by a Hydrogen bond but the bond between two H - O bonds within a water molecule are covalent. The dotted lines represent a hydrogen bond and the solid lines represent a covalent bond.What are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.
What do hydrogen bonds do?
Hydrogen Bonds. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. The attraction created by hydrogen bonds keeps water liquid over a wider range of temperature than is found for any other molecule its size.Is water a covalent bond?
H2O or water as it is more commonly known as is a molecule consisting of 2 Hydrogen molecules bonded to one Oxygen molecule. As the table shows this makes H2O a molecule with a polar covalent bond. Well, electronegativity is the measure of how attracted bond seeking electrons are to an element.What is an example of cohesion?
Cohesion is the term for molecules of a substance sticking together. One of the most common examples is water beading up on a hydrophobic surface. The water will climb up the fibers of the paper, getting it wet above the level of the water in the glass.What are the 4 types of bonds?
4 Types of Chemical Bonds- 1Ionic bond. Ionic bonding involves a transfer of an electron, so one atom gains an electron while one atom loses an electron.
- 2Covalent bond. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms.
- 3Polar bond.