The main reason for adding helium to the breathing mix is to reduce the proportions of nitrogen and oxygen below those of air, to allow the gas mix to be breathed safely on deep dives. Helium has very little narcotic effect. A lower proportion of oxygen reduces the risk of oxygen toxicity on deep dives..
Similarly, you may ask, which gas is used in oxygen cylinder?
Air
Also Know, is there oxygen in helium? Helium has a density that is significantly lower than that of air [4]. Therefore, a mixture of helium and oxygen, termed heliox, has a lower density than does a mixture of nitrogen and oxygen (air, or airox) [6,7].
Also, what does oxygen cylinder contain?
Oxygen cylinders contain oxygen under pressure, and the pressure gauge gradually falls as the cylinder is depleted. A full-size E cylinder (the size fitted to most anaesthetic machines) contains approximately 680 l of gas.
Is helium used in scuba dive tanks?
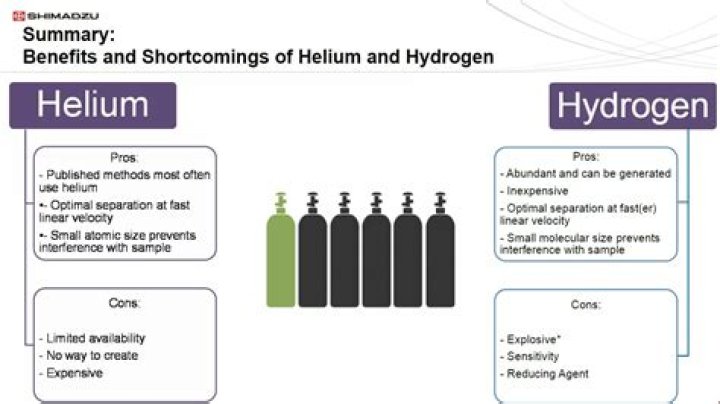
In very deep dives the ratio of oxygen to helium is optimized to even minimize this problem even with oxygen. For normal, no decompression dives, helium is not used in scuba tanks, just normal air( 21% Oxygen and 79% Nitrogen). Technical divers who go beyond recreational depths (30m – 40m) employ the use of trimix.
Related Question Answers
Can we breathe pure oxygen?
We breathe air that is 21 percent oxygen, and we require oxygen to live. So you might think that breathing 100 percent oxygen would be good for us -- but actually it can be harmful. So, the short answer is, pure oxygen is generally bad, and sometimes toxic. When you inhale, the alveoli fill with this air.How does oxygen cylinder work?
When compressed oxygen gas is released from a portable oxygen tank, the high pressure inside the tank forces oxygen outward. The release of pressure creates the oxygen liter flow that travels to your desired oxygen delivery device (such as a nasal cannula or oxygen mask).What gases can we breathe?
Composition. Inhaled air is by volume 78.08% nitrogen, 20.95% oxygen and small amounts of other gasses including argon, carbon dioxide, neon, helium, and hydrogen. The gas exhaled is 4% to 5% by volume of carbon dioxide, about a 100 fold increase over the inhaled amount.Is oxygen a gas?
Oxygen is an element that can be a solid, liquid or gas depending on its temperature and pressure. In the atmosphere it is found as a gas, more specifically, a diatomic gas.What is the weight of a full oxygen cylinder?
Full, it contains 244 cf (6.9 m 3) of oxygen at a pressure of 2200 psi (15200 kPa) as measured at 200C (700F). The oxygen contained in a full cylinder weighs about 20 lb. (9.1 kg); the cylinder itself weighs about 130 lb. (59 kg).Which gas is used in oxygen cylinder in hospital?
For medical centers with a low patient capacity, oxygen is supplied by multiple standard cylinders, as opposed to evaporated liquid oxygen. Nitrous Oxide: Nitrous Oxide, or laughing gas, is used as an analgesic, and as an anesthetic for pre-operative procedures.Why oxygen cylinders are used in hospitals?
Oxygen cylinders are used to transport compressed oxygen gas. They are used in hospitals for artificially administering oxygen to patients. A good ambulance is always equipped with an oxygen cylinder to keep patients breathing while being carried to the hospital.Are oxygen cylinders dangerous?
The dangers are fire and explosion. Oxygen behaves differently to air, compressed air, nitrogen and other inert gases. It is very reactive. Pure oxygen, at high pressure, such as from a cylinder, can react violently with common materials such as oil and grease.How many Litres of oxygen are in a cylinder?
Each vertical unit represents 340 litres, which is equivalent to one standard size cylinder.How thick is an oxygen cylinder?
The thickness varies, being greater at the top and around the bottom but was about 1/4 inch for a medium, old oxygen tank I made something from a few decades ago, IIRC.What is oxygen cylinder valve?
Every oxygen tank or cylinder has a valve that controls the amount of oxygen released. Always open that valve slowly and completely. Here is why: Oxygen molecules are compressed under extreme pressure inside the tank.How many types of oxygen cylinders are there?
You can choose from four main types: Compressed gas system. You'll use this system at home. It includes a stationary oxygen concentrator with a 50-foot tube.Can you breathe helium instead of oxygen?
The bad news is that breathing helium can, in fact, kill you — but not because of the helium, rather because the lack of oxygen when you inhale the helium. As you breathe in a balloon full of helium, you are not breathing in any oxygen, which your cells need – usually we get this from the air we breathe.Can you mix helium and nitrogen?
Helium doesn't mix easily with nitrogen because of the great difference in their densities. But once mixed, the gas molecules are close together and they move around quite a bit with kinetic energy so they stay mixed and don't separate out into layers.What is the difference between oxygen and medical air?
Unlike the other piped medical gases which are typically delivered to hospitals in cylinders, medical air is most often manufactured on-site. Medical air is used for a variety of patient applications. Many patients sensitive to oxygen toxicity are delivered air to lower their exposure to oxygen.What is helium used for?
Helium is commercially recovered from natural gas deposits, mostly from Texas, Oklahoma and Kansas. Helium gas is used to inflate blimps, scientific balloons and party balloons. It is used as an inert shield for arc welding, to pressurize the fuel tanks of liquid fueled rockets and in supersonic windtunnels.Is liquid nitrogen a solution?
Liquid Nitrogen. Nitrogen is a gas. To make it a liquid, it has to be cooled quite extensively. At typical liquid nitrogen temperature of -346°F, it looks like boiling water, even though it's extremely cold.Is oxygen a mixture?
Oxygen is an element and contains no other substances other than oxygen. Thus pure oxygen is not a mixture. Oxygen atoms that we experience in the atmosphere are molecules of O2, two oxygen atoms bonded together. When atoms (elements) bond together they form molecules.