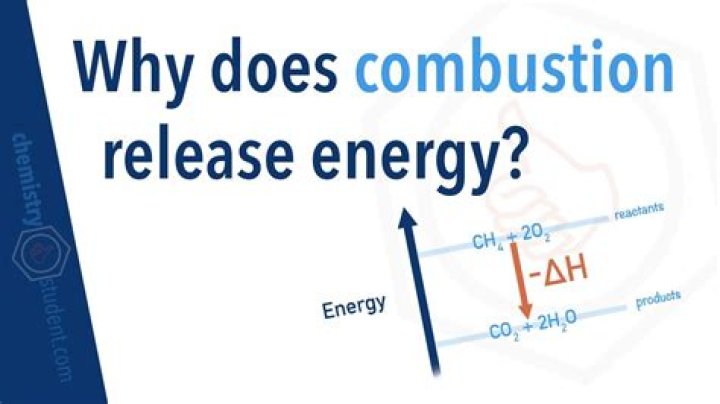
Hydrocarbon fuels like methane (CH4) burn in the presence of oxygen to produce carbon dioxide and water. This process of combustion releases energy. When energy is released during the course of a chemical reaction, it is said to be an EXOTHERMIC reaction..
Considering this, which type of reaction is the burning of gasoline to release heat energy?
Combustion is where you burn a fuel in the presence of an oxidant like oxygen. Heat is produced, because the bonds in the fuel store more energy than the bonds in the water and carbon dioxide that are the products of combustion.
what happens when hydrocarbons burn? Burning hydrocarbons in the presence of oxygen (O2) produces carbon dioxide (Co2) and water (H2O). If there is too much carbon or too little oxygen present when hydrocarbons are burned, carbon monoxide (CO) may also be emitted. Sometimes unburned hydrocarbons are released into the air during incomplete combustion.
Regarding this, what types of energy do combustion reactions release?
Combustion is another name for burning. It is an example of an exothermic reaction, a reaction that releases energy to the surroundings. This is mostly thermal energy, but light energy and sound energy are also released.
Is boiling endothermic or exothermic?
We can all appreciate that water does not spontaneously boil at room temperature; instead we must heat it. Because we must add heat, boiling water is a process that chemists call endothermic. Clearly, if some processes require heat, others must give off heat when they take place. These are known as exothermic.
Related Question Answers
How much energy is released in combustion of octane?
The heat of combustion of octane (C8H18, Mm = 114 g/mol) is -5500 kJ/mol.What does heat of combustion mean?
Heat of combustion (ΔH°c) is the measure of the amount of energy released in the form of heat (q) when one mole of a substance is burned (combustion). The production of heat means the reaction is an exothermic process and gives off energy.What happens to chemical bonds when fuel burns?
Combustion (burning) is essentially a chemical reaction between a fuel and oxygen. Atoms in the fuel are held together by chemical bonds. When heat is added to fuel and oxygen, the atoms in the fuel are energized and start to break apart from one another. Once released, these atoms form new bonds with the oxygen.What are the 2 types of combustion?
1. Complete combustion: - Complete combustion: During complete combustion, the reactant burns in oxygen and produces products that are limited.
- Incomplete Combustion:
- Smoldering:
- Rapid Combustion:
- Spontaneous combustion:
- Turbulent combustion:
- Micro-combustion:
What are the three types of combustion?
The three important types of combustion are: - Rapid combustion.
- Spontaneous combustion.
- Explosive combustion.
How much heat does it release on burning?
The heat of combustion is approximately -418 kJ per mole of O2 used up in the combustion reaction, and can be estimated from the elemental composition of the fuel.What type of energy is wood burning?
chemical energy
Is Rusting a fast or a slow reaction?
A: Rusting can happen quickly or slowly, depending on the material that's rusting, and the environment. Rust is the oxidation of iron along with the absorption of water to make Fe2O3 with water molecules attached.What is the definition of activation energy in chemistry?
The term Activation Energy was introduced in 1889 by Svante Arrhenius, a Swedish scientist. It is defined as the least possible amount of energy (minimum) which is required to start a reaction or the amount of energy available in a chemical system for a reaction to take place.Do all combustion reactions release energy?
Hydrocarbon fuels like methane (CH4) burn in the presence of oxygen to produce carbon dioxide and water. This process of combustion releases energy. When energy is released during the course of a chemical reaction, it is said to be an EXOTHERMIC reaction.What type of reaction is combustion?
A combustion reaction is a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat. Combustion reactions must involve O2 as one reactant. The combustion of hydrogen gas produces water vapor (see figure below).How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What is energy of combustion?
Chemistry and Chemical Technology The heat of combustion (energy content) of natural gas is the amount of energy that is obtained from the burning of a volume of natural gas, measured in British thermal units (Btu). The value of natural gas is calculated by its Btu content.What happens when fuels are burned?
Complete combustion Fuels are substances that react with oxygen to release useful energy. Most of the energy is released as heat, but light energy is also released. When they burn completely: the carbon oxidises to carbon dioxide.How does octane combust?
2 C8H18 + 25 O2 ---> 16 CO2 +18 H2O. Combustion reactions are classified as rapid reactions that produce a flame. This is because lower octane gas ignites at lower temperatures and is as such more flammable. Higher octane gas requires more compression and higher temperatures to burn efficiently.How do you tell if it's complete or incomplete combustion?
Combustion requires three things to occur: fuel (hydrocarbons), oxygen (from the air), and a catalytic spark. Complete combustion will produce only carbon dioxide and water as the products and nothing will be leftover. Incomplete combustion will produce other byproducts like carbon monoxide or carbon soot left behind.Are all hydrocarbons flammable?
Many hydrocarbons are highly flammable; therefore, care should be taken to prevent injury. If hydrocarbons undergo combustion in tight areas, toxic carbon monoxide can form.What are the 3 products of oxygen when it has been burned?
Description. Regardless of the type of hydrocarbon, combustion with oxygen produces 3 products: carbon dioxide, water and heat, as shown in the general reaction below.Is coal a hydrocarbon?
Hydrocarbon resource. Hydrocarbon resources are resources that contain hydrocarbon molecules which means it consists both hydrogen and carbon. Hydrocarbon resources are often known as fossil fuels (natural gas, oil, and coal) since hydrocarbons are the primary constituent in these.